7977. Pyridostigmine Bromide
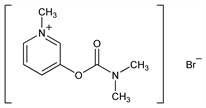
Nomenclature
Description and references
Reversible inhibitor of acetylcholinesterase. Prepn: Urban, US 2572579 (1951 to Hoffmann-La Roche). Mechanism of protective effect in soman poisoning: X. Deyi et al., Fundam. Appl. Toxicol. 1, 217 (1981). Evaluation of side effects profile under desert conditions: J. E. Cook et al., Mil. Med. 157, 250 (1992). Comparative clinical trial for reversal of neuromuscular block: L. Gyermek et al., Br. J. Anaesth. 74, 410 (1995). Review of determn methods in biological samples: B. Zhao et al., J. Pharm. Pharm. Sci. 9, 71-81 (2006).
Properties
Shiny, hygroscopic crystals from abs ethanol, mp 152-154°. Characteristic, agreeable odor. Freely sol in water, alcohol, chloroform; slightly sol in hexane. Practically insol in ether, acetone, benzene. Aq solns may be sterilized by autoclaving with steam.Therapeutic Category
Cholinergic; in treatment of myasthenia gravis. Reversal agent for neuromuscular blocking drugs. Pre-exposure antidote to chemical warfare agents.
Therapeutic Category (Veterinary)
In treatment of myasthenia gravis.Keywords
Cholinergic; Antimyasthenic; Neuromuscular Block Reversant; Cholinesterase Inhibitor