7980. Pyridoxamine Dihydrochloride
Nomenclature
CAS number: 524-36-7
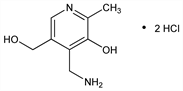
4-(Aminomethyl)-5-hydroxy-6-methyl-3-pyridinemethanol dihydrochloride; 2-methyl-3-hydroxy-4-aminomethyl-5-hydroxymethylpyridine dihydrochloride. C8H14Cl2N2O2; mol wt 241.11.
C 39.85%, H 5.85%, Cl 29.41%, N 11.62%, O 13.27%.
Description and references
Synthesis and structure: Harris et al., J. Biol. Chem. 154, 315 (1944); J. Am. Chem. Soc. 66, 2088 (1944).
Properties
Platelets, mp 226-227° with dec. Soluble in water (approx 1 g/2 ml); sol in 95% alc (0.65 g/100 ml). Reasonably stable at room temp; shows no decompn in a few days at 60°. Liquefies on exposure to 80% relative humidity. pH of a 1% water soln 2.4. uv max (pH 1.94): 287.5 nm (E mol 91,000).Derivative
Free base.Properties
Crystals from alc; mp 193-193.5°; sol in alc.Derivative
Pyridoxamine phosphate.Nomenclature
2-Methyl-3-hydroxy-4-aminomethyl-5-pyridylmethylphosphoric acid.C8H13N2O5P; mol wt 248.17.
C 38.72%, H 5.28%, N 11.29%, O 32.23%, P 12.48%.
Description and references
Prepn in soln by autoclaving codecarboxylase with glutamic acid: McNutt, Snell, J. Biol. Chem. 182, 557 (1950); prepn by direct phosphorylation of pyridoxamine in aq soln with phosphorus oxychloride: Heyl et al., J. Am. Chem. Soc. 73, 3430 (1951).