7981. 4-Pyridoxic Acid
Nomenclature
CAS number: 82-82-6
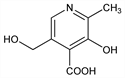
3-Hydroxy-5-(hydroxymethyl)-2-methyl-4-pyridinecarboxylic acid; 3-hydroxy-5-(hydroxymethyl)-2-methylisonicotinic acid; 2-methyl-3-hydroxy-4-carboxy-5-hydroxymethylpyridine. C8H9NO4; mol wt 183.16.
C 52.46%, H 4.95%, N 7.65%, O 34.94%.
Description and references
Occurs in urine. It is the chief metabolic product of pyridoxine, pyridoxal, and pyridoxamine. Isoln from human urine: Singal, Sydenstricker, Science 78, 545 (1941). Isoln and synthesis: Huff, Perlzweig, J. Biol. Chem. 155, 345 (1944).
Properties
Wedge-shaped crystals, mp 247-248°. Slightly sol in water, alcohol, pyridine. Insoluble in ether and in aq acid soln, but completely sol in aq alkaline soln. Possesses two acidic groups, one a phenolic and the other a carboxyl having pK values of 9.75 and 5.50, respectively. Characteristic blue fluorescence, max at pH 3 to 4. The fluorescence disappears on reduction with hydrosulfite and is restored to the original intensity with H2O2. Is adsorbed on zeolite from aq solns at pH 4 to 5 and can be eluted with 25% KCl; butanol extracts of neutral eluates also show characteristic blue fluorescence which is increased by a trace of acetic acid. Stable to boiling with dil alkali (1N); but upon being heated with 0.5N acid for a few minutes it is converted to the lactone.Derivative
Lactone.Nomenclature
β-Pyracine.C8H7NO3; mol wt 165.15.
C 58.18%, H 4.27%, N 8.48%, O 29.06%.