7984. Pyrilamine
Nomenclature
CAS number: 91-84-9
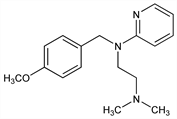
N-[(4-Methoxyphenyl)methyl]-N′,N′-dimethyl-N-2-pyridinyl-1,2-ethanediamine; 2-[(2-dimethylaminoethyl)(p-methoxybenzyl)amino]pyridine; N-p-methoxybenzyl-N′,N′-dimethyl-N-α-pyridylethylenediamine; mepyramine; pyranisamine; RP-2786. C17H23N3O; mol wt 285.38.
C 71.55%, H 8.12%, N 14.72%, O 5.61%.
Description and references
Prepn: Bovet et al., C. R. Seances Soc. Biol. Ses Fil. 138, 99 (1944); Huttrer et al., J. Am. Chem. Soc. 68, 1999 (1946); Viaud, Prod. Pharm. 2, 53 (1947); Horclois, US 2502151 (1950 to Rh^one-Poulenc). Toxicity: A. von Schlichtergroll, Arzneim.-Forsch. 7, 237 (1957); of the maleate: F. Hunziker et al., ibid. 13, 324 (1963).
Properties
Oily liquid. bp5 201°; bp0.06 168-172°. nD25 1.5760-1.5765. LD50 orally in mice: 312 mg/kg (Schlichtergroll).Derivative
Hydrochloride.C17H23N3O.HCl; mol wt 321.84.
C 63.44%, H 7.52%, N 13.06%, O 4.97%, Cl 11.02%.
Properties
Crystals, mp 143-143.5°. Very sol in water.Derivative
Maleate.Nomenclature
CAS number: 59-33-6
Antamine (Grove); Anthisan (M & B); Dorantamin (Dorsey); Enrumay (Cooper); Histalon (Hance); Histan (Cooper); Histapyran (Horton & Converse); Histatex (Medix); Neo-Antergan (Poulenc); Paraminyl (Cooper); Parmal (Central); Pyramal (Roxane); Stamine (Tutag); Stangen (Phys. Drug); Thylogen (Rorer). Properties
Crystals. Bitter saline taste. Stable in air. mp 100-101°. uv max: 244 nm (E1%1cm 420). One gram dissolves in about 0.4 ml water, in about 15 ml abs alc. pH of 10% soln ≈5.1. On raising the pH to 7.5 or 8.0 pptn of the oily, free base begins. Slightly sol in benzene, ether. LD50 orally in mice: 338 mg/kg (Hunziker).Therapeutic Category
Antihistaminic.
Therapeutic Category (Veterinary)
Antihistaminic.Keywords
Antihistaminic; Ethylenediamine Derivatives