7985. Pyrimethamine
Nomenclature
Description and references
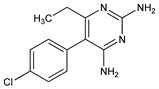
Dihydrofolate reductase inhibitor; generally used in combination with other antimicrobial agents. Prepn: P. B. Russell, G. H. Hitchings, J. Am. Chem. Soc. 73, 3763 (1951); G. H. Hitchings et al., US 2576939 (1951 to Burroughs Wellcome); W. Logemann et al., Ber. 87, 435 (1954); R. M. Jacob, US 2680740 (1954 to Rh^one-Poulenc). Review of antimicrobial activity and mechanism of action: Burchall in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 312-320. Comprehensive description: M. A. Loutfy, H. Y. Aboul-Enein, Anal. Profiles Drug Subs. 12, 463-482 (1983). LC-MS determn in plasma: B. A. Sinnaeve et al., J. Chromatogr. A 1076, 97 (2005). Clinical evaluations in toxoplasmosis in AIDS patients: C. Leport et al., Am. J. Med. 84, 94 (1988); B. Dannemann et al., Ann. Intern. Med. 116, 33 (1992). Review of clinical experience in malaria: H. M. McIntosh et al., Ann. Trop. Med. Parasitol. 93, 265-270 (1998); C. V. Plowe et al., Br. Med. J. 328, 545 (2004).
Properties
Crystals, mp 233-234° (capillary); mp 240-242° (copper block). Practically insol in water. Slightly sol in ethanol, (about 9 g/l), in dil HCl (about 5 g/l); sol in boiling ethanol (about 25 g/l). Very sparingly sol in propylene glycol and dimethylacetamide at 70°.Derivative
Combination with sulfadoxine.Nomenclature
Therapeutic Category
Antiprotozoal (Toxoplasma); antimalarial.
Therapeutic Category (Veterinary)
Antiprotozoal (Toxoplasma).Keywords
Antimalarial; Antiprotozoal (Toxoplasma)