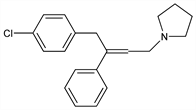
8012. Pyrrobutamine
Nomenclature
CAS number: 91-82-7
1-[4-(4-Chlorophenyl)-3-phenyl-2-butenyl]pyrrolidine; 1-[γ-p-(chlorobenzyl)cinnamyl]pyrrolidine; 1-p-chlorophenyl-2-phenyl-4-pyrrolidyl-2-butene; Pyronil (Lilly). C20H22ClN; mol wt 311.85.
C 77.03%, H 7.11%, Cl 11.37%, N 4.49%.
Description and references
Prepn: J. Mills, US 2655509 (1953 to Eli Lilly). Pharmacology and toxicity: H. M. Lee et al., Proc. Soc. Exp. Biol. Med. 80, 458 (1952). Pharmacology and physical properties: N. G. Lordi, J. E. Christian, J. Am. Pharm. Assoc. 45, 300 (1956). Structure-activity and stereochemical study: A. F. Casy, R. R. Ison, J. Pharm. Pharmacol. 22, 270 (1970). GC-MS determn in urine: H. Maurer, K. Pfleger, J. Chromatogr. 430, 31 (1988).
Properties
Oily liquid, bp0.3 190-195°. On standing gives crystals, mp 48-49°. d25 1.1052. uv max (95% ethanol): 360, 243 nm (α 112, 9500), (c = 0.538, 0.00538 mg/ml).Derivative
Diphosphate.Nomenclature
CAS number: 135-31-9
C20H28ClNO8P2; mol wt 507.84.
C 47.30%, H 5.56%, Cl 6.98%, N 2.76%, O 25.20%, P 12.20%.
Properties
Crystals from alcohol + ether, mp 129.5-130°. pKa1 8.77, pka2 5.23. Sol in warm water to the extent of 10%. Soly in alcohol at 25° about 5%. Soly at pH 7.4 (37.5°): 0.00087 M/l. Practically insol in chloroform, ether. LD50 in mice, guinea pigs (mg/kg): 1270 ±156, 1241 ±165 s.c.; 836.8 ±95, 625.6 ±41.9 i.m.; 1116 ±73, 992.6 ±107 orally; in mice (mg/kg): 53.53 ±1.61 i.v. (Lee).Derivative
Hydrochloride.C20H22ClN.HCl; mol wt 348.31.
C 68.97%, H 6.66%, Cl 20.36%, N 4.02%.
Properties
Crystals from alcohol + ether, mp 227-228°.Derivative
Hydrobromide.C20H22ClN.HBr; mol wt 392.76.
C 61.16%, H 5.90%, Cl 9.03%, N 3.57%, Br 20.34%.
Properties
Crystals from alcohol + ether, mp 228-229°.Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic; Alkylamine Derivatives