8033. Quercetagetin
Nomenclature
CAS number: 90-18-6
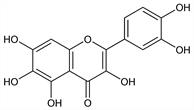
2-(3,4-Dihydroxyphenyl)-3,5,6,7-tetrahydroxy-4H-1-benzopyran-4-one; 3,3′,4′,5,6,7-hexahydroxyflavone; 6-hydroxycyanidenolon 1555. C15H10O8; mol wt 318.24.
C 56.61%, H 3.17%, O 40.22%.
Description and references
From flowers of French marigold, Tagetes patula Linn., Compositae: Perkin, J. Chem. Soc. 103, 209 (1913). Synthesis: Baker et al., ibid. 1929, 74; Rao, Seshadri, Proc. Indian Acad. Sci. 23A, 23 (1946), C.A. 40, 50522 (1946).
Derivative
Dihydrate.Properties
Pale yellow needles from dil alcohol, mp 318°. uv max (alc): 259, 361 nm (log ε 4.23, 4.34). Sol in hot alcohol; sparingly sol in boiling water.Derivative
Hexaacetate.C27H22O14; mol wt 570.46.
C 56.85%, H 3.89%, O 39.27%.
Properties
Needles from alcohol + acetic acid, mp 209-211°. Sparingly sol in alc.Derivative
7-Glucoside.Nomenclature
Quercetagitrin.C21H20O13; mol wt 480.38.
C 52.51%, H 4.20%, O 43.30%.
Description and references
From flowers of the African marigold, Tagetes erecta L., Compositae: Rao, Seshadri, Proc. Indian Acad. Sci. 14A, 289 (1941), C.A. 36, 25553 (1942); from Chrysanthemum coronarium L., Compositae: Anyas, Steelink, Arch. Biochem. Biophys. 90, 63 (1960). Structure: Rajagopalan, Seshadri, Proc. Indian Acad. Sci. 28A, 31 (1948), C.A. 43, 4265b (1949).