8034. Quercetin
Nomenclature
Description and references
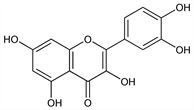
The aglucon of quercitrin, of rutin, and of other glycosides. Widely distributed in the plant kingdom, esp in rinds and barks, in clover blossoms and in ragweed pollen. Isoln from Rhododendron cinnabarinum Hook, Ericaceae: Rangaswami et al., Proc. Indian Acad. Sci. 56A, 239 (1962), C.A. 58, 9414a (1963). Structure: Underhill et al., Can. J. Biochem. Physiol. 35, 219 (1957). Biosynthesis: Watkin et al., ibid. 229; Grisebach, Biochem. J. 85, 3p (1962); Patschke et al., Z. Naturforsch. 21b, 201 (1966). Synthesis: Shakhova et al., Zh. Obshch. Khim. 32, 390 (1962), C.A. 58, 1426f (1963). Metabolism: Nakagawa et al., Biochim. Biophys. Acta 97, 233 (1965). Toxicity data: M. Sullivan et al., Proc. Soc. Exp. Biol. Med. 77, 269 (1951). See also Bioflavonoids.
Derivative
Dihydrate.Nomenclature
Properties
Yellow needles from dil alcohol. Becomes anhydr at 95-97°. When anhydr dec 314°. uv max (alc): 258, 375 nm (log ε 2.75, 2.75). One gram dissolves in 290 ml abs alc, in 23 ml boiling alc. Soluble in glacial acetic acid; in aq alkaline solns with yellow color. Practically insol in water. Alcoholic solns taste very bitter. LD50 orally in mice: 160 mg/kg (Sullivan).Derivative
Pentabenzyl ether.Nomenclature
Description and references
Prepn: Chopin, Chadenson, C. R. Seances Acad. Sci. Ser. C 263, 729 (1966); Binovic, DE 2122514 (1972 to Biosedra), C.A. 76, 113072n (1972).
Properties
Crystals, mp 123-125°. uv max (chloroform): 249, 343 nm (log ε 4.43, 4.14).Derivative
3-β-d-Galactoside hemipentahydrate.Nomenclature
Hyperin; hyperoside.Description and references
From Acacia melanoxylon R. Br., Leguminosae: Falco, de Vries, Naturwissenschaften 51, 462 (1964).
Properties
Yellow needles from ethanol, dec 227-230°. [α]D20 -83° (c = 0.2 in pyridine). uv max: 259, 364 nm (log ε 4.31, 4.39).Therapeutic Category
Capillary protectant.
Keywords
Vasoprotectant