8047. Quinaldine
Nomenclature
Description and references
Occurs in coal tar; made from aniline, acetaldehyde and HCl. Lab procedure: A. I. Vogel, Practical Organic Chemistry (Longmans, London, 3rd ed., 1959) p 831; Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 318. Pharmacology: Brown et al., Comp. Biochem. Physiol. 42A, 223 (1972). Toxicity data: Smyth et al., Arch. Ind. Hyg. Occup. Med. 4, 119 (1951); L. L. Marking, Invest. Fish Control 23, 3 (1969).
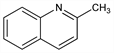
Properties
Colorless, oily liquid; quinoline odor; becomes reddish-brown on exposure to air. d ≈1.06. bp 246-247°. Practically insol in water. Sol in chloroform, ether. Keep tightly closed and protected from light. LD50 orally in rats: 1.23 g/kg (Smyth).Derivative
Sulfate.Nomenclature
Description and references
Prepn: S. Hoogewerff, W. A. Van Dorp, Rec. Trav. Chim. 3, 345 (1884); J. L. Allen, J. B. Sills, Invest. Fish Control 47, 3 (1973). Efficacy as anesthetic in fish: P. A. Gilderhus et al., ibid. 49, 3 (1973); G. C. Blasiola Jr., J. Fish Biol. 10, 113 (1977). Toxicity: L. L. Marking, V. K. Dawson, Invest. Fish Control 48, 3 (1973).