8060. Quinidine
Nomenclature
Description and references
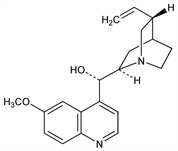
A dextrorotatory stereoisomer of quinine, q.v. Present in cinchona barks to the extent of 0.25-3.0%. Found in quinine sulfate mother liquors. Review of structural elucidation and early synthetic studies: R. B. Turner, R. B. Woodward, in The Alkaloids vol. 3, 1-63 (1953). Configuration: Prelog, Zalán, Helv. Chim. Acta 27, 535 (1944); Prelog, H"afliger, ibid. 33, 2021 (1950); Roth, Pharmazie 16, 257 (1961). Crystal and molecular structure: R. Doherty et al., J. Pharm. Sci. 67, 1698 (1978). Rotatory dispersion studies: Lyle, Gaffield, Tetrahedron Lett. 1963, 1371. Prepn by isomerization of quinine: W. E. Doering et al., J. Am. Chem. Soc. 69, 1700 (1949). Total synthesis: J. Gutzwiller, M. Uskokovic, ibid. 92, 204 (1970); eidem, Helv. Chim. Acta 56, 1494 (1973); eidem, J. Am. Chem. Soc. 100, 576 (1978). Toxicity data: C. Turba et al., Arzneim.-Forsch. 18, 1127 (1968); K. Dietmann et al., ibid. 27, 589 (1977). Comprehensive description of the sulfate: M. A. Loutfy et al., Anal. Profiles Drug Subs. 12, 483-546 (1983). Clinical evaluation in severe malaria: R. E. Phillips et al., N. Engl. J. Med. 312, 1273 (1985). Review of pharmacology and clinical efficacy in cardiac arrhythmias: J. W. Mason, L. M. Hondeghem, Ann. N.Y. Acad. Sci. 432, 162-176 (1984); A. R. Leon, J. D. Merlino, Heart Dis. Stroke 2, 407-413 (1993).
Properties
Triboluminescent. mp 174-175° after drying of solvated crystals. [α]D15 +230° (c = 1.8 in chloroform), [α]D17 +258° (alc), [α]D17 +322° (c = 1.6 in 2M HCl). pK1 (20°) 5.4; pK2 10.0. Blue fluorescence in dil H2SO4. The uv absorption spectrum is identical with that of quinine. One gram dissolves in about 2000 ml cold, 800 ml boiling water, 36 ml alcohol, 56 ml ether, 1.6 ml chloroform; very sol in methanol. Practically insol in petr ether. LD50 in rats (mg/kg): 30 i.v., 263 orally (Dietmann).Derivative
Hemipentahydrate.Properties
Prisms from dil alcohol, loses 1/2 H2O in air, mp ≈168°.Derivative
Hydrogen sulfate tetrahydrate.Nomenclature
Properties
Rods, sol in 8 parts water with blue fluorescence.Derivative
Sulfate dihydrate.Nomenclature
Properties
White, very bitter, odorless, fine crystals, frequently cohering in masses. Darkens on exposure to light. Does not lose all of its water below 120°. [α]D25 ≈+212° (95% alcohol); ≈+260° (dil HCl). pKa 4.2, 8.8. pH (1% aq soln): 6.0-6.8. One gram dissolves in about 90 ml water, 15 ml boiling water, 10 ml alcohol, 3 ml methanol, 12 ml chloroform. Insol in ether, benzene. Protect from light. LD50 in mice, rats (mg/kg): 700, 455.8 orally; 83, 56 i.v. (Turba).Derivative
Gluconate.Nomenclature
Properties
Crystals, mp 175-176.5°. Sol in 9 parts water, 60 parts alcohol.Derivative
Polygalacturonate.Nomenclature
Description and references
(C20H24N2O2.C6H10O7.H2O)x. Prepn: A. Halpern et al., Am. J. Pharm. 130, 190 (1958). Pharmacology: A. Halpern et al., Antibiot. Chemother. 9, 97 (1959).
Properties
Amorphous powder, mp 180° (dec). Anhydr product is insol in methanol, ethanol, chloroform, ether, acetone, dioxane. Soly in hot 40% methanol or ethanol: 12%; in water at 25°: ≈2%. LD50 in rats, mice (mg/kg): 3200 ± 350, 2680 ± 210 orally (Halpern, 1959).Therapeutic Category
Antiarrhythmic (class IA); antimalarial.
Therapeutic Category (Veterinary)
Antiarrhythmic.Keywords
Antiarrhythmic; Antimalarial