8061. Quinine
Nomenclature
Description and references
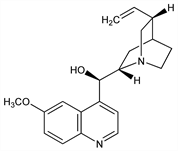
Primary alkaloid of various species of Cinchona (Rubiaceae), see Cinchona. Representative samples of dried bark contain ≈0.8 to 4% quinine. Optical isomer of quinidine, q.v. Isoln: Pelletier, Caventau, Ann. Chim. Phys. [2] 15, 291 (1820). Extraction procedure: Jucker, Stoll, in Ullmanns Encyklop"adie der technischen Chemie vol. 3, 213-218 (1953). Configuration: Prelog, Zalán, Helv. Chim. Acta 27, 535 (1944); Prelog, H"afliger, ibid. 33, 2021 (1950); Roth, Pharmazie 16, 257 (1961). Synthesis: Woodward, Doering, J. Am. Chem. Soc. 66, 849 (1944); 67, 860 (1945); Taylor, Martin, ibid. 94, 6218 (1972); Gutzwiller, Uskokovic, ibid. 100, 576 (1978); G. Grethe et al., ibid. 589; T. Imanishi et al., Chem. Pharm. Bull. 30, 1925 (1982). Review of structural elucidation and early synthetic studies: R. B. Turner, R. B. Woodward in The Alkaloids vol. 3, 1-63 (1953); of bioactivity: F. E. Hahn, Ed. in Antibiotics vol. 5 (pt. 2) (Springer-Verlag, New York, 1979) pp 353-362. Comprehensive description of the hydrochloride: F. J. Muhtadi et al., Anal. Profiles Drug Subs. 12, 547-621 (1983). LC determn in soft drinks: L. P. Valenti, J. Assoc. Off. Anal. Chem. 68, 782 (1985). HPLC determn in blood: V. K. Dua et al. J. Chromatogr. 614, 87 (1993). Clinical evaluation to relieve nocturnal leg cramps: P. S. Connolly et al., Arch. Intern. Med. 152, 1877 (1992). Clinical efficacy in malaria: P. G. Kremsner et al., J. Infect. Dis. 169, 467-470 (1994). Review of historical importance and total syntheses: T. S. Kaufman, E. A. Ruveda, Angew. Chem. Int. Ed. 44, 854-885 (2005).
Properties
Triboluminescent, orthorhombic needles from abs alcohol, mp 177° (some decompn). Sublimes in high vacuum at 170-180°. [α]D15 -169° (c = 2 in 97% alcohol), [α]D17 -117° (c = 1.5 in chloroform), [α]D15 -285° (c = 0.4M in 0.1N H2SO4). pK1 (18°) 5.07; pK2 9.7. pH of satd aq soln 8.8. Absorption spectra: Dobbie, Lauder, J. Chem. Soc. 99, 1260 (1911); Dobbie, Fox, ibid. 101, 78 (1912). Fluorescence: Rabe, Marschall, Ann. 382, 362 (1911). The blue fluorescence is especially strong in dil H2SO4. One gram dissolves in 1900 ml water, 760 ml boiling water, 0.8 ml alcohol, 80 ml benzene (in 18 ml benzene at 50°), in 1.2 ml chloroform; 250 ml dry ether, 20 ml glycerol, 1900 ml of 10% ammonia water. Almost insol in petr ether.Derivative
Trihydrate.Properties
Microcrystalline powder, mp 57°, efflorescent, loses one H2O in air, two H2O over H2SO4, anhydr at 125°.Derivative
Bisulfate heptahydrate.Nomenclature
Properties
Very bitter crystals or cryst powder; efflorescent on exposure to air and darkens on exposure to light. One gram dissolves in 9 ml water, 0.7 ml boiling water, 23 ml alcohol, 0.7 ml alcohol at 60°, 625 ml chloroform, 2500 ml ether, 15 ml glycerol. pH: 3.5.Derivative
Dihydrochloride.Nomenclature
Properties
Very bitter powder or crystals. One gram dissolves in about 0.6 ml water, in about 12 ml alcohol. Slightly sol in chloroform, very slightly sol in ether. Aq solns are strongly acid to litmus paper (pH about 2.6).Derivative
Hydrochloride dihydrate.Nomenclature
Properties
Bitter, silky needles. Effloresces on exposure to warm air. Does not lose all its water below 120°. One gram dissolves in 16 ml water, in 0.5 ml boiling water, in 1.0 ml alcohol, in about 7.0 ml glycerol, in about 1 ml chloroform, in about 350 ml ether. pH (1% aq soln): 6.0-7.0. Bitterness threshold 1:30000. Protect from light.Derivative
Sulfate dihydrate.Nomenclature
Properties
Dull needles or rods, making a light and readily compressible mass. Becomes brownish on exposure to light. Loses its water of crystn at about 100°. [α]D15 -220° (5% soln in about 0.5N HCl). One gram dissolves in 810 ml water, 32 ml boiling water, 120 ml alcohol, 10 ml alcohol at 78°. Slightly sol in chloroform, ether, but freely sol in a mixture of 2 vols chloroform and 1 vol abs alcohol. Aq solns are neutral to litmus, pH of satd soln 6.2.Use
Flavor in carbonated beverages.Therapeutic Category
Antimalarial; muscle relaxant (skeletal).
Therapeutic Category (Veterinary)
Antiprotozoal for fish.Keywords
Antimalarial; Muscle Relaxant (Skeletal)