Nomenclature
CAS number: 106-51-4
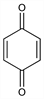
2,5-Cyclohexadiene-1,4-dione;
p-quinone; 1,4-benzoquinone; 1,4-cyclohexadienedione.
C
6H
4O
2; mol wt 108.09.
C 66.67%, H 3.73%, O 29.60%.
Description and references
Made by oxidation of aniline with sodium dichromate
in presence of sulfuric acid. Laboratory prepn from hydroquinone:
Vliet, Org. Synth. 2, 85 (1922); also Org. Synth. coll. vol. I (2nd ed., 1941). Cf. Underwood, Walsh, ibid. 16, 73 (1936). Toxicity
study: Woodard et al., Fed. Proc. 8, 348 (1949).
Properties
Yellow monoclinic prisms from water or petr ether.
Penetrating odor resembling that of chlorine. Irritating vapors.
d420 1.318. mp 115.7°. Sublimes [sublimation velocities in vacuo: Kempf, J. Prakt. Chem. [2] 78, 236 (1908)]. Volatile with steam. Absorption spectrum: Hantzsch, Ber. 49, 522 (1916). Dipole moment:
0.67. Polemic over correct values: Paoloni, J. Am. Chem. Soc. 80, 3879 (1958). Slightly
sol in water; sol in alcohol, ether, hot petr ether, alkalies. LD50 orally in rats:
130 mg/kg (Woodard).Caution
Potential symptoms of overexposure
are eye and skin irritation, conjunctivitis; keratitis. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 272.Use
Oxidizing agent; in photography; manuf dyes; manuf
hydroquinone; tanning hides; making gelatin insol; strengthening animal
fibers; as reagent.
