8104. Ramoplanin
Nomenclature
Description and references
Glycolipodepsipeptide antibiotic complex consisting of 6 related components (A1, A2, A3, A′1, A′2, A′3) of which A2 is the most abundant. Isoln and inital characterization from Actinoplanes sp.: B. Cavalleri et al., DE 3013246; eidem, US 4303646 (1980, 1981 both to Gruppo Lepetit); idem et al., J. Antibiot. 37, 309 (1984). Bactericidal activity: R. Pallanza et al., ibid. 318. Structure elucidation: R. Ciabatti et al., ibid. 42, 254 (1989); and sequence determn: J. K. Kettenring et al., ibid. 268. 3D structure: M. Kurz, W. Guba, Biochemistry 35, 12570 (1996). Isoln and characterization of the A′ factors: L. Gastaldo et al., J. Ind. Microbiol. 11, 13 (1992). Determn in sera: M. T. Kenny et al., Diagn. Microbiol. Infect. Dis. 18, 117 (1994). Clinical evaluation against vancomycin resistant enterococci (VRE): M. T. Wong et al., Clin. Infect. Dis. 33, 1476 (2001). Review of discovery and chemistry: F. Parenti et al., Drugs Exp. Clin. Res. 16, 451-455 (1990); of chemistry and mechanism of action: S. Walker et al., Chem. Rev. 105, 449-475 (2005); of pharmacology and clinical potential: D. K. Farver et al., Ann. Pharmacother. 39, 863-868 (2005).
Properties
White powder, slightly hygroscopic. mp 210-230°. [α]D20 +78.3° (c = 1.04 in H2O). Sol in DMF and lower alcohols. Insol in ethyl ether, petroleum ether and benzene. LD50 in mice (mg/kg): 328 i.p., 122 i.v.; orally in rats: 2000 mg/kg (Pallanza).Derivative
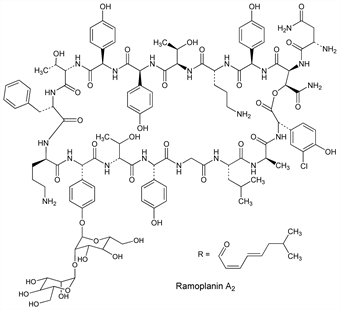
Ramoplanin A2.Nomenclature
Description and references
Total synthesis: W. Jiang et al., J. Am. Chem. Soc. 124, 5288 (2002); eidem, ibid. 125, 1877 (2003).
Properties
White powder, mp 210-220°. [α]D20 +73±4° (c = 0.49 in H2O). uv max in methanol: 234, 268 nm (E1cm1% 206, 114).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Polypeptides