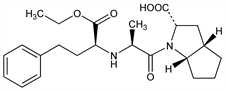
8103. Ramipril
Nomenclature
Description and references
Angiotensin converting enzyme (ACE) inhibitor; converted to active, diacid metabolite, ramiprilat. Prepn: V. Teetz et al, EP 79022 (1983 to Hoechst A.G.), C.A. 100, 52012h (1984); E. H. Gold et al., US 4587258 (1986 to Schering Corp.); V. Teetz et al., Arzneim.-Forsch. 34, 1399 (1984). Radioimmunoassay determn in human serum and plasma: H. G. Eckert et al., ibid. 35, 1251 (1985). Toxicology: H. H. Donaubauer, D. Mayer, ibid. 38, 14 (1988). Symposium on pharmacology and clinical efficacy: Am. J. Cardiol. 59, 1D-177D (1987). Clinical trial in chronic nephropathy: P. Ruggenenti et al., Lancet 352, 1252 (1998). Clinical prevention of cardiovascular events in high-risk patients: HOPE Study Investigators, N. Engl. J. Med. 342, 145 (2000). Clinical trial in prevention of stroke: J. Bosch et al., Br. Med. J. 324, 1 (2002).
Properties
Felty needles from ether, mp 109°. [α]D24 +33.2° (c = 1 in 0.1N ethanolic HCl). LD50 (14 day) in male, female mice, male, female rats (mg/kg): 1194, 1158, 687, 608 i.v.; 10933, 10048, >10000, >10000 orally (Donaubauer, Mayer).Therapeutic Category
Antihypertensive.
Keywords
ACE-Inhibitor; Antihypertensive; N-Carboxyalkyl (peptide/lactam) Derivatives