8171. Rhamnetin
Nomenclature
CAS number: 90-19-7
2-(3,4-Dihydroxyphenyl)-3,5-dihydroxy-7-methoxy-4H-1-benzopyran-4-one; 3,3′,4′,5-tetrahydroxy-7-methoxyflavone; 7-methylquercetin; β-rhamnocitrin; cyanidenolon-7-methyl ether 1537. C16H12O7; mol wt 316.26.
C 60.76%, H 3.82%, O 35.41%.
Description and references
The aglucon of xanthorhamnin. From fruit of Rhamnus cathartica L., Rhamnaceae: Krassowski, J. Russ. Phys. Chem. Soc. 40, 1510 (1909); from commercial xanthorhamnin: Nystrom et al., J. Org. Chem. 22, 1272 (1957). Structure: Oesch, Perkin, J. Chem. Soc. 105, 2354 (1914); Jurd, Horowitz, J. Org. Chem. 22, 1618 (1957). Synthesis: Kuhn, Low, Ber. 77, 211 (1944); Jurd, J. Am. Chem. Soc. 80, 5531 (1958); US 2892845 (1959 to USDA); Anand et al., J. Sci. Ind. Res. 21B, 322 (1962), C.A. 57, 13712f (1962); Kawano et al., Chem. Pharm. Bull. 15, 711 (1967).
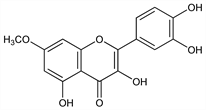
Properties
Yellow needles from acetone + methanol, mp 292-294°. uv max (ethanol): 371, 256 nm (log ε 4.41, 4.40). Sol in hot phenol. Slightly sol in hot water, hot alcohol, hot glacial acetic acid, hot acetone. Freely sol in dil alkalies with intense yellow color.Derivative
Tetraacetate.C24H20O11; mol wt 484.41.
C 59.51%, H 4.16%, O 36.33%.