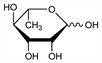
8172. Rhamnose
Nomenclature
CAS number: 3615-41-6
6-Deoxy-l-mannose; l-rhamnose; l-mannomethylose; isodulcit. C6H12O5; mol wt 164.16.
C 43.90%, H 7.37%, O 48.73%.
Description and references
Occurs free in poison sumac (Rhus toxicodendron L., Anacardiaceae), combined in the form of glycosides of many plants. Preparation: Clark, J. Biol. Chem. 38, 255 (1919). Structure and configuration: Fischer, Morrell, Ber. 27, 384 (1894); Fischer, Zach, ibid. 45, 3762 (1912); Hirst, Macbeth, J. Chem. Soc. 1926, 23; Avery, Hirst, ibid. 1929, 2466. Isoln from walls of gram-negative bacteria: Salton, Biochim. Biophys. Acta 45, 364 (1960); from Afraegle paniculata Engl., Rutaceae: Torto, J. Chem. Soc. 1961, 5234; from rabbit skin: Malawista, Davidson, Nature 192, 871 (1961); from leaves of Solanum chacoense Bitter, Solanaceae: Kuhn, L"ow, Ber. 94, 1088 (1961).