8190. Rhodomycins
Description and references
Antibiotic substance produced by Streptomyces purpurascens nov. sp.: Brockmann, Bauer, Naturwissenschaften 37, 492 (1950); Brockmann et al., Ber. 84, 700 (1951); GB 708749 (1954 to Bayer). Separation of rhodomycin A and rhodomycin B: Brockmann, Patt, Ber. 88, 1455 (1955). Isoln and structural elucidation of additional rhodomycins produced by Streptomyces stammes: Biedermann, Brauniger, Pharmazie 27, 782 (1972); Brockmann et al., Tetrahedron Lett. 1973, 3699. Total synthesis of racemic rhodomycinones (the aglycones of the rhodomycins): A. S. Kende, Y.-G. Tsay, Chem. Commun. 1977, 140.
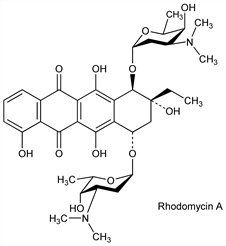
Derivative
Rhodomycin A.Nomenclature
Description and references
Dec on mild acidic hydrolysis into 1 mole β-rhodomycinone and 2 moles rhodosamine: Brockmann, Spohler, Naturwissenschaften 48, 716 (1961). Structure of β-rhodomycinone: Brockmann, Niemeyer, ibid. 570. Structure of rhodosamine: Brockmann, Spohler, ibid. 42, 154 (1955). Total structure: Brockmann et al., Tetrahedron Lett. 1969, 415.
Derivative
Dihydrochloride.Properties
Red prisms from ethanol + isopropanol, mp 205°. Freely sol in water, lower alcohols; sparingly sol in benzene, chloroform; practically insol in ether, petr ether.Derivative
Perchlorate.Properties
Minute red needles, mp 188°. Freely sol in methanol, acetone; sparingly sol in water, butanol.Derivative
Rhodomycin B.Nomenclature
Description and references
Dec on mild acidic hydrolysis into 1 mole β-rhodomycinone and 1 mole rhodosamine: Brockmann, Spohler, ibid. 48, 716 (1961).