8200. Riboflavin
Nomenclature
Description and references
Nutritional factor found in milk, eggs, malted barley, liver, kidney, heart, leafy vegetables. Richest natural source is yeast. Minute amounts present in all plant and animal cells. Occurs in the free form only in the retina of the eye, in whey, and in urine. Bioactive forms occurring in tissues and cells are riboflavin monophosphate and flavine-adenine dinucleotide, q.q.v. Syntheses: Karrer et al., Helv. Chim. Acta 18, 426, 522 (1935); Kuhn et al., Naturwissenschaften 23, 260 (1935); Howe, US 2807611 (1957 to Merck & Co.); F. Yoneda et al., J. Chem. Soc. Perkin Trans. 1 1978, 348. May also be produced by fermentation: Malzahn et al., US 2876169 (1959 to Grain Processing Corp.). Toxicity studies: Kuhn, Boulanger, Z. Physiol. Chem. 241, 233 (1936); K. Unna, J. G. Greslin, J. Pharmacol. Exp. Ther. 76, 75 (1942). Early reviews: Rosenberg in The Vitamins (New York, 1945); Sebrell, Harris, The Vitamins vol. III (Academic Press, New York, 1954) pp 299-402; Vogel-Knobloch, Chemie und Technik der Vitamine vol. II, part 1 (Enke, Stuttgart, 1955). Review of metabolism: Rivlin, N. Engl. J. Med. 283, 463 (1970). Review of extraction and assay methods: Pearson, The Vitamins vol. VII, P. Gy"orgy, W. N. Pearson, Eds. (Academic Press, New York, 2nd ed., 1967) pp 99-136. Comprehensive reviews: ibid. vol. V, pp 1-96 (1972); F. J. Al-Shammary et al., Anal. Profiles Drug Subs. 19, 429-476 (1990).
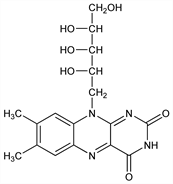
Properties
Fine orange-yellow needles from 2N acetic acid, alcohol, water, or pyridine. Dec at 278-282° (darkens at about 240°). Three different crystal forms having different solubilities in water: Dale, US 2603633; US 2797215. [α]D25 -112 to -122° (50 mg in 2 ml 0.1N alcoholic NaOH dil to 10 ml with water). Absorption max: 220-225, 266, 371, 444, 475 nm. One gram dissolves in from 3000 to about 15,000 ml of water, the variations in the soly being due to differences in the crystal structure. Soly in alcohol (27.5°): 0.0045 g/100 ml of abs ethanol. Slightly sol in cyclohexanol, amyl acetate, benzyl alcohol, phenol; very sol in dil alkalies (with decompn). Insol in ether, chloroform, acetone, benzene. pKa 10.2; pKb 1.7. pH of satd aq soln: ≈6. Isoelectric pt: pH 6. Aq solns are yellow showing a green fluoresence with max at 565 nm. Sensitive to alkalies; stable to mineral acids in the dark. Visible or ultraviolet irradiation of alkaline solns causes the formation of lumiflavine, q.v., whereas irradiation of acid or neutral solns gives rise to the production of lumichrome, q.v., together with varying amounts of lumiflavine. LD50 in rats (g/kg): >10 orally, 5.0 s.c., 0.56 i.p. (Unna, Greslin).Derivative
2′,3′,4′,5′-Tetrabutyrate.Nomenclature
Therapeutic Category
Vitamin (enzyme cofactor).
Therapeutic Category (Veterinary)
Vitamin (enzyme cofactor).Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B2