8211. Ricinoleic Acid
Nomenclature
CAS number: 141-22-0
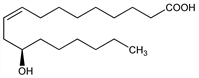
(9Z,12R)-12-Hydroxy-9-octadecenoic acid; d-12-hydroxyoleic acid. C18H34O3; mol wt 298.46.
C 72.44%, H 11.48%, O 16.08%.
Description and references
Found primarily in oils from the seeds of Ricinus spp, Euphorbiaceae. Accounts for about 90% of the triglyceride fatty acids of castor oil, and up to about 40% of the glyceride fatty acids of ergot oil. Bibliography on its isoln: Ralston, Fatty Acids (New York, 1948) p 189. Also isolated from Linum mucronatum (flax), Linaceae: Kleiman, Spencer, Lipids 6, 962 (1971). Structure: Goldsobel, Ber. 27, 3121 (1894). Mechanism of biosynthesis: Morris, Biochem. Biophys. Res. Commun. 29, 311 (1967).
Properties
Liquid. d427.4 0.940; mp 5.5°; bp10 245°. [α]D22 +6.67°; [α]D26 +7.15° (c = 5 in acetone). nD20 1.4716. Neutralization value 187.98; iodine value 85.05. Sol in alcohol, acetone, ether, chloroform (cf. the solubilities of castor oil).Derivative
Acid sulfate.Nomenclature
CAS number: 36634-48-7
Ricinolsulfuric acid. C18H34O6S; mol wt 378.52.
C 57.12%, H 9.05%, O 25.36%, S 8.47%.
Properties
Obtained by the action of chlorosulfonic acid. Viscous brown liquid with weak blue fluorescence. Sol in water (about 10%), alcohol, ether, chloroform.Derivative
Sodium salt.Nomenclature
CAS number: 5323-95-5
Sodium ricinoleate; Soricin; Colidosan. C18H33NaO3; mol wt 320.44.
C 67.47%, H 10.38%, Na 7.17%, O 14.98%.
Properties
Sodium salts of the fatty acids from castor oil. White or slightly yellow, odorless or almost odorless powder. Sol in water or alcohol. The aq soln is alkaline.Use
In textile finishing; sometimes added to Turkey red oil, dry-cleaning soaps.Therapeutic Category
Has been used in contraceptive jellies. The sodium salt has been used as sclerosing agent.
Keywords
Sodium Ricinoleate: Sclerosing Agent