8217. Rifamycins
Nomenclature
Rifomycins.Description and references
Group of antibiotics characterized by a natural ansa structure (chromophoric naphthohydroquinone group spanned by a long aliphatic bridge) not previously found in other known antibiotics. Isoln from the fermentation broths of Streptomyces mediterranei: P. Sensi et al., Antibiot. Annu. 1959-1960, 262. Among rifamycins, rifamycin B, O, S, SV and X are the more studied members. Prepn of rifamycin B derivs: P. Sensi et al., US 3313804 (1967 to Lepetit). Structure: Prelog, Pure Appl. Chem. 7, 551 (1963); Chemotherapia 7, 133 (1963); Oppolzer et al., Experientia 20, 336 (1964); Oppolzer, Prelog, Helv. Chim. Acta 56, 2287 (1973). Total synthesis of rifamycin S: H. Nagaoka et al., J. Am. Chem. Soc. 102, 7962 (1980); H. Iio et al., ibid. 7965; H. Nagaoka, Y. Kishi, Tetrahedron 37, 3873 (1981); S. Hanessian et al., J. Am. Chem. Soc. 104, 6164 (1982). Review of chemistry of ansamycin antibiotics: K. L. Rinehart et al., Fortschr. Chem. Org. Naturst. 33, 231-307 (1976). General reviews: P. Sensi, Research Progress in Organic-Biological & Medicinal Chemistry vol. 1, U. Gallo, L. Santamaria, Eds. (Società Editoriale Farmaceutica, Milan, Italy, 1964) pp 337-421; Wehrli, Staehelin, in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 252-268; C. Gurgo et al., “Rifamycins” in Handbook of Experimental Pharmacology vol. 61, G. V. R. Born et al., Eds., entitled “Chemotherapy of Viral Infections”, P. E. Came, L. A. Caliguiri, Eds. (Springer-Verlag, New York, 1982) pp 519-555.
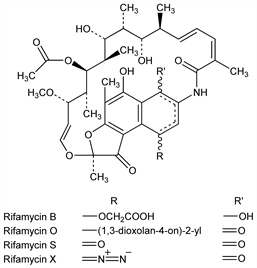
Derivative
Rifamycin AMP see Rifampin.Derivative
Rifamycin B.Nomenclature
Properties
Yellow prismatic needles from benzene, mp 300° (dec 160-164°). [α]D20 -11° (methanol). Absorption max (phosphate buffer soln pH 7.3): 223, 304, 425 nm (E1%1cm 555, 275, 220). Dibasic acid. Very stable. Solubilities: water 0.027% (w/w), methanol 2.62%, ethanol 0.44%. LD50 in mice (mg/kg): 2040 i.v., >3000 i.p., s.c., and orally (Sensi, 1964).Derivative
Rifamycin O.Nomenclature
Description and references
Prepn: P. Sensi et al., Farmaco Ed. Sci. 15, 228 (1960); Umezawa, JP 66 15518 (1966), C.A. 66, 1583v (1967).
Properties
Pale yellow crystals from methanol, mp 300° (dec 160°). Also reported as mp 180-185° (Umezawa). [α]D20 +71.5° (c = 1 in dioxane). uv max (methanol contg 5% acetate buffer soln pH 4.62): 226, 273, 370 nm (E1%1cm 365, 440, 60). Weak acid. Slowly sol in alkaline soln with red-violet color. Sol in acetone, tetrahydrofuran; slightly sol in methanol, ethanol, ethyl acetate. Practically insol in ether, petr ether, water, dilute acids.Derivative
Rifamycin S.Nomenclature
Description and references
Activation product found in solns of rifamycin B and rifamycin O: P. Sensi et al., Experientia 16, 412 (1960).
Properties
Yellow-orange crystals from methanol, dec 179-181°. [α]D20 +476° (c = 0.1 in methanol). Absorption max (phosphate buffer soln pH 7.3): 317, 525 nm (E1%1cm 426, 62). LD50 in mice (mg/kg): 122 i.v.; 258 i.p.; 3000 orally (Sensi, 1964).Derivative
Rifamycin SV see separate entry.Derivative
Rifamycin X.Description and references
Prepn: Greco et al., Farmaco Ed. Sci. 16, 766 (1961).
Properties
Yellow crystals, no definite mp, dec 135-140°. Unstable to light. [α]D20 +491.8° (c = 0.981 in dioxane). Absorption max (acetate buffer soln): 286, 317, 402 nm (E1%1cm 400, 362, 195). Practically insol in water. Sol in methanol, ethanol, ethyl acetate, benzene.Derivative
Rifamycin AG.Description and references
A condensation product of rifamycin O and aminoguanidine: P. Sensi et al., Antibiot. Chemother. 12, 448 (1962).