8218. Rifamycin SV
Nomenclature
CAS number: 6998-60-3
5,6,9,17,19,21-Hexahydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione 21-acetate; rifomycin SV; rifamicine SV. C37H47NO12; mol wt 697.77.
C 63.69%, H 6.79%, N 2.01%, O 27.52%.
Description and references
Semi-synthetic antibiotic derived from Rifamycin S. Prepn: Sensi et al., Experientia 16, 412 (1960); Farmaco Ed. Sci. 16, 165 (1961). Comprehensive review: Bergamini, Fowst, Arzneim.-Forsch. 15, 951-1002 (1965). For general refs see Rifamycins.
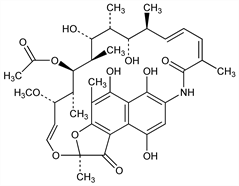
Properties
Yellow-orange crystals, mp 300° (dec 140°). [α]D20 -4° (methanol). uv max (phosphate buffer soln pH 7.3): 223, 314, 445 nm (E1%1cm 586, 322, 204). Acid reaction. Slightly sol in water, petr ether; sol in ether, bicarbonate soln; very sol in methanol, ethanol, acetone, ethyl acetate. A reducing substitute, such as ascorbic acid, should be added to aq solns of rifamycin SV to prevent its transformation to rifamycin S. LD50 in mice (mg/kg): 550 i.v.; 625 i.p.; 2120 orally (Bergamini, Fowst).Derivative
Sodium salt.Nomenclature
CAS number: 14897-39-3
Rifamastene (Uce); Rifocin (Lepetit). Properties
Orange-red crystals. Soly in water pH 7.2: ≈5 g/100 ml.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Ansamycins