8232. Risedronic Acid
Nomenclature
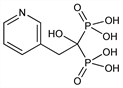
CAS number: 105462-24-6
[1-Hydroxy-2-(3-pyridinyl)ethylidene]bisphosphonic acid; 2-(3-pyridinyl)-1-hydroxyethane diphosphonic acid. C7H11NO7P2; mol wt 283.11.
C 29.70%, H 3.92%, N 4.95%, O 39.56%, P 21.88%.
Description and references
Bisphosphonate antiresorptive agent. Prepn: J. J. Benedict, C. M. Perkins, EP 186405; eidem, US 5583122 (1986, 1996 both to Procter & Gamble). Pharmacology: W. S. S. Jee et al., Bone 14, 493 (1993). Clinical trial in Paget's disease: E. S. Siris et al., J. Bone Miner. Res. 13, 1032 (1998); in postmenopausal bone loss: L. Mortensen et al., J. Clin. Endocrinol. Metab. 83, 396 (1998). Clinical effect on reduction of fracture risk: S. T. Harris et al., J. Am. Med. Assoc. 282, 1344 (1999). Review of pharmacology and clinical efficacy: K. L. Goa, J. A. Balfour, Drugs Aging 13, 83-91 (1998).
Derivative
Monosodium salt.Nomenclature
CAS number: 115436-72-1
Risedronate sodium; NE-58095; Actonel (Procter & Gamble); Optinate (Procter & Gamble). C7H10NNaO7P2; mol wt 305.09.
C 27.56%, H 3.30%, N 4.59%, Na 7.54%, O 36.71%, P 20.30%.
Properties
Fine, white to off-white, odorless crystalline powder. Occurs as the hemi-pentahydrate. Sol in water. Essentially insol in common organic solvents.Therapeutic Category
Bone resorption inhibitor.
Keywords
Antiosteoporotic; Antipagetic; Bone Resorption Inhibitor