8236. Ritipenem
Nomenclature
CAS number: 84845-57-8
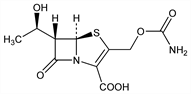
(5R,6S)-3-[[(Aminocarbonyl)oxy]methyl]-6-[(1R)-1-hydroxyethyl]-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic
acid; (5R,6S,8R)-6α-hydroxyethyl-2-carbamoyloxymethyl-2-penem-3-carboxylic
acid; (5R,6S)-6-[(1R)-1-hydroxyethyl]-3-(hydroxymethyl)-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic
acid, 3-carbamate. C10H12N2O6S; mol wt 288.28.
C 41.66%, H 4.20%, N 9.72%, O 33.30%, S 11.12%.
Description and references
Prepn: M. Alpegiani et al., DE 3245270; M. Foglio et al., US 4482565 (1983, 1984 both to Carlo Erba). Synthesis: G. Franceschi et al., J. Antibiot. 36, 938 (1983). Total synthesis: W. Cabri et al., Tetrahedron Lett. 34, 3491 (1993). Toxicity study: M. Brughera et al., J. Antimicrob. Chemother. 23, Suppl. C, 129 (1989). Series of articles on synthesis, in vitro activity, metabolism: ibid. 1-204 (1989). Clinical pharmacokinetics of acid and ester forms: S. R. Norrby et al., ibid. 25, 371 (1990); A. M. Lovering et al., ibid. 29, 179 (1992). HPLC determn in serum and urine: R. Mendez et al., J. Chromatogr. 579, 115 (1992).
Derivative
Sodium salt.Nomenclature
CAS number: 84845-58-9
FCE-22101. C10H11N2NaO6S; mol wt 310.26.
C 38.71%, H 3.57%, N 9.03%, Na 7.41%, O 30.94%, S 10.33%.
Properties
[α]D20 +140°. uv max (H2O): 258, 306 nm (ε 4150, 6030). LD50 in male, female mice, male, female rats (mg/kg): 3872, 4393, 2000, 2201 i.v. (Brughera).Derivative
Acetoxymethyl ester.Nomenclature
CAS number: 87238-52-6
Ritipenem acoxil; FCE-22891. C13H16N2O8S; mol wt 360.34.
C 43.33%, H 4.48%, N 7.77%, O 35.52%, S 8.90%.
Properties
LD50 in male, female mice, male, female rats (mg/kg): 4363, 6167, >5000, >5000 orally (Brughera).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penems