8237. Ritodrine
Nomenclature
CAS number: 26652-09-5
(αS)-rel-4-Hydroxy-α-[(1R)-1-[[2-(4-hydroxyphenyl)ethyl]amino]ethyl]benzenemethanol; erythro-p-hydroxy-α-[1-[(p-hydroxyphenethyl)amino]ethyl]benzyl
alcohol; N-[2-(p-hydroxyphenyl)ethyl]-N-[2-(p-hydroxyphenyl)-2-hydroxy-1-methylethyl]amine; 1-(4-hydroxyphenyl)-2-[2-(4-hydroxyphenyl)ethylamino]propanol; N-(p-hydroxyphenylethyl)-4-hydroxynorephedrine. C17H21NO3; mol wt 287.35.
C 71.06%, H 7.37%, N 4.87%, O 16.70%.
Description and references
β2-Adrenergic agonist. Prepn: BE 660244 (1965 to N.V. Philips); Claassen et al., US 3410944 (1968 to No. Am. Philips). Clinical investigations: Coutinho et al., Am. J. Obstet. Gynecol. 104, 1053 (1969); Landesman et al., ibid. 110, 111 (1971); Wesselius-De Casparis et al., Br. Med. J. 3, 144 (1971). Clinical efficacy in treatment of preterm labor: J. F. Larsen et al., Obstet. Gynecol. 67, 607 (1986).
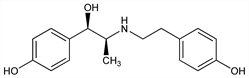
Derivative
Base.Properties
Resinous mass, mp 88-90°.Derivative
Hydrochloride.Nomenclature
CAS number: 23239-51-2
DU-21220; Miolene (Lusofarmaco); Prempar (Duphar); Pre-Par (Duphar); Utemerin (Kissei); Utopar (Ferrosan); Yutopar (Astra). C17H21NO3.HCl; mol wt 323.81.
C 63.06%, H 6.85%, N 4.33%, O 14.82%, Cl 10.95%.
Properties
mp 193-195° (dec) from ethanol-ether. uv max: 267.5 nm (ε 3310).Therapeutic Category
Tocolytic.
Keywords
β-Adrenergic Agonist; Tocolytic