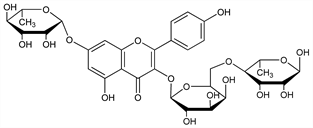
8243. Robinin
Nomenclature
CAS number: 301-19-9
3-[[6-O-(6-Deoxy-α-l-mannopyranosyl)-β-d-galactopyranosyl]oxy]-7-[(6-deoxy-α-l-mannopyranosyl)oxy]-5-hydroxy-2-(4-hydroxyphenyl)-4H-1-benzopyran-4-one; kaempferol 3-robinoside 7-rhamnoside. C33H40O19; mol wt 740.66.
C 53.51%, H 5.44%, O 41.04%.
Description and references
Dimorphic flavanoid isolated from the leaves and flowers of Robinia pseudoacacia L., Leguminosae: C. Zwenger, F. Dronke, Ann. suppl. 1, 257 (1861); C. Sando, J. Biol. Chem. 94, 675 (1932). Structure: Zemplén, Bognár, Ber. 74B, 1783 (1941). Total synthesis and structure: L. Farkas et al., Phytochemistry 15, 215 (1976).