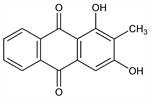
8281. Rubiadin
Nomenclature
CAS number: 117-02-2
1,3-Dihydroxy-2-methyl-9,10-anthracenedione; 1,3-dihydroxy-2-methylanthraquinone. C15H10O4; mol wt 254.24.
C 70.86%, H 3.96%, O 25.17%.
Description and references
From Rubia tinctorum L., Coprosma var., Morinda citrifolia Linn, Rubiaceae: Schunck, Ann. 87, 344 (1853); Briggs, Nicholls, J. Chem. Soc. 1949, 1241; Briggs et al., ibid. 1952, 1718; Borvie, Cooke, Aust. J. Chem. 15, 332 (1962). Structure: Marchlewski, J. Chem. Soc. 63, 1137 (1893); Schunck, Marchlewski, ibid. 65, 182 (1894); Jones, Robertson, ibid. 1930, 1699. Synthesis: Joshi et al., J. Sci. Ind. Res. 14B, 87 (1955); Hirase, Chem. Pharm. Bull. 8, 417 (1960).