8318. SAICAR
Nomenclature
CAS number: 3031-95-6
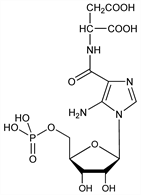
N-[[5-Amino-1-(5-O-phosphono-β-d-ribofuranosyl)-1H-imidazol-4-yl]carbonyl]-l-aspartic
acid; N-[(5-amino-1-ribofuranosylimidazol-4-yl)carbonyl]aspartic
acid 5′-phosphate; 5-amino-N-(1,2-dicarboxyethyl)-1-ribofuranosylimidazole-4-carboxamide
5′-phosphate; 5-amino-4-imidazole-N-succinocarboxamide ribonucleotide; 5-amino-4-imidazole-N-succinocarboxamide ribotide; N-[5-amino-1-(5′-phosphoribofuranosyl)-4-imidazolecarbonyl]aspartic
acid; succino-AICAR. C13H19N4O12P; mol wt 454.28.
C 34.37%, H 4.22%, N 12.33%, O 42.26%, P 6.82%.
Description and references
Important purine precursor. Synthesis by enzymic reaction according to the equation AIR + ATP + aspartic acid + CO2 → SAICAR + ADP + orthophosphate: Lukens, Buchanan, J. Biol. Chem. 234, 1791 (1959). Production by adenine-requiring mutants of Bacillus subtilis: Ohmura et al., US 3280007 (1966 to Takeda). Synthesis of SAICAR and derivatives: Burrows, Shaw, J. Chem. Soc. C 1967, 1088; Burrows et al., ibid. 1968, 40.
Derivative
Dibarium salt.C13H15Ba2N4O12P; mol wt 724.91.
C 21.54%, H 2.09%, Ba 37.89%, N 7.73%, O 26.49%, P 4.27%.