8319. Sakuranetin
Nomenclature
CAS number: 2957-21-3
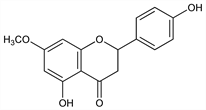
2,3-Dihydro-5-hydroxy-2-(4-hydroxyphenyl)-7-methoxy-4H-1-benzopyran-4-one; 4′,5-dihydroxy-7-methoxyflavanone; naringen 7-methyl ether. C16H14O5; mol wt 286.28.
C 67.13%, H 4.93%, O 27.94%.
Description and references
Isoln by hydrolysis of 5-glucoside obtained from bark of Prunus yedoensis Matsum., Rosaceae: Asahina, Arch. Pharm. 246, 259 (1908); from wood of various Prunus species: Hasegawa, Shirato, J. Am. Chem. Soc. 76, 5559 (1954); 77, 3557 (1955); Hasegawa, ibid. 79, 1738 (1957). Structure: Asahina et al., J. Pharm. Soc. Jpn. No. 550, 1007 (1927); Shinoda, Sato, ibid. 48, 220 (1928). Synthesis: Zemplén et al., Ber. 75B, 1432 (1942). Absolute configuration of (+)-isomer: Arakawa, Nakazaki, Ann. 636, 111 (1960).
Properties
Needles, mp 152°. Also reported as dihydrate, mp 131-132° (Arakawa, Nakazaki, loc. cit.). (+)-Isomer, [α]D16 +8.0° (c = 7.92 in acetone); [α]D12 +8.4° (c = 6.28 in methanol). Sol in alcohol, ether, chloroform, benzene, ethyl acetate, pyridine; slightly sol in boiling water. Practically insol in cold water.Derivative
5-Glucoside.Nomenclature
4′,5-dihydroxy-7-methoxyflavanone 5-glucoside; sakuranin.C22H24O10; mol wt 448.42.
C 58.93%, H 5.39%, O 35.68%.