8324. Salicin
Nomenclature
CAS number: 138-52-3
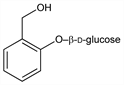
2-(Hydroxymethyl)phenyl-β-d-glucopyranoside; salicoside; salicyl alcohol glucoside; saligenin-β-d-glucopyranoside. C13H18O7; mol wt 286.28.
C 54.54%, H 6.34%, O 39.12%.
Description and references
Usually obtained by making hot water extracts from the ground bark of poplar (Populus) and willow (Salix); also found in the leaves and female flowers of the willow. Isoln from root bark of Viburnum prunifolium L., Caprifoliaceae: Evans et al., J. Am. Pharm. Assoc. 34, 207 (1945). Structure and synthesis: Irvine, Rose, J. Chem. Soc. 89, 814 (1906); Kunz, J. Am. Chem. Soc. 48, 262 (1926). Hydrolysis by acids: Moelwyn-Hughes, Trans. Faraday Soc. 25, 503 (1929). Enzymatic hydrolysis: Pigman, J. Res. Natl. Bur. Stand. 27, 6 (1941). Enzymatic hydrolysis in heavy water: Stacie, Z. Phys. Chem. 28B, 236 (1935).
Properties
Orthorhombic crystals from water, mp 199-202°. [α]D25 -62 to -67° (c = 3). [α]D20 -45.6° (c = 0.6 in abs alc), Brauns, J. Am. Chem. Soc. 47, 1292 (1925). One gram dissolves in 23 ml water, 3 ml boiling water, 90 ml alcohol, 30 ml alcohol at 60°. Soluble in alkalies, pyridine, glacial acetic acid. Practically insol in ether, chloroform. Aq solns are neutral to litmus and have a bitter taste.Use
Standard substrate in evaluating enzyme prepns contg β-glucosidase: Weidenhagen, Z. Ver. Zucker-Ind. 79, 591 (1929).Therapeutic Category
Analgesic.
Therapeutic Category (Veterinary)
Has been used as a bitter stomachic, as an antirheumatic and as an analgesic.Keywords
Analgesic; Non-opioids; Salicylic Acid Derivatives