8332. Salicylic Acid
Nomenclature
Description and references
Occurs in the form of esters in several plants, notably in wintergreen leaves and the bark of sweet birch. Manuf by heating sodium phenolate with carbon dioxide under pressure: W. L. Faith et al., Industrial Chemicals (Wiley, New York, 3rd ed., 1965) pp 652-655. Review of prepns, properties, and uses of salicylic acid and derivatives: S. H. Erickson in Kirk-Othmer Encyclopedia of Chemical Technology vol. 20 (John Wiley & Sons, New York, 3rd ed., 1982) pp. 500-524. Toxicity data: K. Sota et al., J. Pharm. Soc. Jpn. 89, 1392 (1969). HPLC determn in foods: B. H. Chen, S. C. Fu, Chromatographia 41, 43 (1995). Clinical evaluation to treat warts: B. J. Bart et al., J. Am. Acad. Dermatol. 20, 74 (1989); M. E. Bender et al., Cutis 47, 199 (1991). Review of use in acne: E. Zander, S. Weisman, Clin. Ther. 14, 247 (1992). Comprehensive description: M. A. Abounassif et al., Anal. Profiles Drug Subs. Excip. 23, 421-470 (1994).
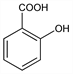
Properties
White crystals, fine needles, or fluffy white crystalline powder, mp 159°; sublimes at 76°. Gradually discolors in sunlight. d420 1.443. bp20 211°. pKa 2.98. n 1.565. Flash point (closed cup): 157°C. uv max (4 mg% in ethanol): 210, 234, 303 nm (ε 8343, 5466, 3591). Soly (Wt %): water 0.20 (20°), 2.21 (80°); carbon tetrachloride 0.262 (25°); benzene 0.775 (25°); propanol 27.36 (21°); abs ethanol 34.87 (21°); acetone 396 (23°). pH of satd aq soln 2.4. Log P (octanol/water) 2.188 ±0.0548. Keep protected from light. LD50 i.v. in mice: 500 mg/kg (Sota).Derivative
Sodium salt.Nomenclature
Description and references
Clinical trial in rheumatoid arthritis: S. J. Preston et al., Br. J. Clin. Pharmacol. 27, 607 (1989). Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971).
Properties
White crystalline powder, mp 440°. Soly (g/100 cc): water 125; methanol 17. Protect from light. LD50 i.p. in rats: 780 mg/kg (Goldenthal).Caution
Ingestion of large amounts can cause vomiting, abdominal pain, increased respiration, acidosis, mental disturbances. May cause skin rashes in sensitive individuals. See: Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984) pp 368-375.Use
In manuf aspirin, methyl salicylate, and other salicylates. Coupling agent for azo dyes. Has been used as food preservative. Buffer.Therapeutic Category
Keratolytic. Sodium salt as anti-inflammatory, analgesic, antipyretic.
Therapeutic Category (Veterinary)
Keratolytic.Keywords
Keratolytic
Sodium Salicylate: Anti-inflammatory (Nonsteroidal); Salicylic Acid Derivatives; Antipyretic; Analgesic; Non-opioids; Salicylic Acid Derivatives