8344. Salvinorins
Nomenclature
Divinorins.Description and references
Family of neoclerodane diterpenes isolated from the hallucinogenic Mexican mint Salvia divinorum. Traditional medicine used by the Mazatec people of Oaxaca, Mexico. Isoln of Salvinorin A: A. Ortega et al., J. Chem. Soc. Perkin Trans. 1 1982, 2505; of A and B and biologic activity: L. J. Valdés, III et al., J. Org. Chem. 49, 4716 (1984); of C: L. J. Valdés, III et al., Org. Lett. 3, 3935 (2001). Stereochemistry: M. Koreeda et al., Chem. Lett. 1990, 2015. Pharmacologic effects: D. J. Siebert, J. Ethnopharmacol. 43, 53 (1994). Review: L. J. Valdés III, J. Psychoactive Drugs 26, 277-283 (1994).
Derivative
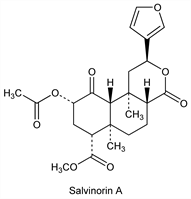
Salvinorin A.Nomenclature
Description and references
One of the most potent natural hallucinogens known. First naturally occurring non-nitrogenous κ opioid receptor (KOR) agonist identified. HPLC quantification in S. divinorum extracts: J. W. Gruber et al., Phytochem. Anal. 10, 22 (1999). LC determn in biological fluids: M. S. Schmidt et al., J. Chromatogr. B 818, 221 (2005). KOR binding profile: B. L. Roth et al., Proc. Natl. Acad. Sci. USA 99, 11934 (2002). Brief review: D. J. Sheffler, B. L. Roth, Trends Pharmacol. Sci. 24, 107-109 (2003).