8368. Sapropterin
Nomenclature
Description and references
Natural cofactor of the aromatic amino acid hydroxylases required for catecholamine and serotonin biosynthesis. Identification of cofactor activity: S. Kaufman, Proc. Natl. Acad. Sci. USA 50, 1085 (1963). Prepn of (6R,S)-BH4: B. Schircks et al., Helv. Chim. Acta 61, 2731 (1978). Chromatographic separation of diastereoisomers: S. W. Bailey, J. E. Ayling, J. Biol. Chem. 253, 1598 (1978). Absolute configuration of natural isomer: W. L. F. Armarego et al., Aust. J. Chem. 35, 785 (1982). Stereospecific synthesis: S. Matsuura et al., Heterocycles 23, 3115 (1985); H. Sakai, T. Kanai, EP 191335; eidem, US 4713454 (1986, 1987 both to Shiratori; Suntory). Bioavailability: G. Kapatos, S. Kaufman, Science 212, 955 (1981). Effect on neurotransmitter monoamine biosynthesis: S. Miwa et al., Arch. Biochem. Biophys. 239, 234 (1985). LC determn in biological samples: Y. Tani, T. Ishihara, Life Sci. 46, 373 (1990). Therapeutic potential in hyperphenylalaninemia: S. Kaufman, J. Nutr. Sci. Vitaminol, Suppl., 601 (1992). Clinical trial in phenylketonuria: H. L. Levy et al., Lancet 370, 504 (2007). Review of development and clinical experience: J. R. Burnett, IDrugs 10, 805-813 (2007).
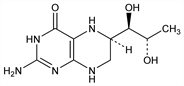
Properties
pK′ 5.05. uv max (0.1 N HCl): 265 nm (ε 14000).Derivative
Dihydrochloride.Nomenclature
Properties
Crystals from HCl, mp 245-246° (dec). [α]D25 -6.81° (c = 0.665 in 0.1 M HCl). uv max (2 M HCl): 264 nm (ε 16770).Therapeutic Category
In treatment of hyperphenylalaninemia.
Keywords
Enzyme Cofactor