8369. Saquinavir
Nomenclature
CAS number: 127779-20-8
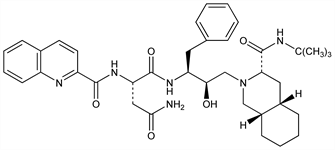
(2S)-N1[(1S,2R)-3-[(3S,4aS,8aS)-3-[[(1,1-Dimethylethyl)amino]carbonyl]octahydro-2(1H)-isoquinolinyl]-2-hydroxy-1-(phenylmethyl)propyl]-2-[(2-quinolinylcarbonyl)amino]butanediamide; (S)-N-[(αS)-α-[(1R)-2-[(3S,4aS,8aS)-3-(tert-butylcarbamoyl)octahydro-2(1H)-isoquinolyl]-1-hydroxyethyl]phenethyl]-2-quinaldamido
succinamide; N-tert-butyldecahydro-2-[2(R)-hydroxy-4-phenyl-3(S)-[[N-(2-quinolylcarbonyl)-l-asparaginyl]amino]butyl](4aS,8aS)-isoquinoline-3(S)-carboxamide; Ro-31-8959. C38H50N6O5; mol wt 670.84.
C 68.04%, H 7.51%, N 12.53%, O 11.92%.
Description and references
Peptidomimetic HIV protease inhibitor. Prepn: J. A. Martin, S. Redshaw, EP 432695; eidem, US 5196438 (1991, 1993 both to Hoffmann-LaRoche); K. E. B. Parkes et al., J. Org. Chem. 59, 3656 (1994). In vitro HIV proteinase inhibition: N. A. Roberts et al., Science 248, 358 (1990). Antiviral properties: J. C. Craig et al., Antiviral Res. 16, 295 (1991); S. Galpin et al., Antivir. Chem. Chemother. 5, 43-45 (1994). Clinical evaluation of tolerability and activity: V. S. Kitchen et al., Lancet 345, 952 (1995). Review of pharmacology and clinical experience: S. Kravcik, Expert Opin. Pharmacother. 2 303-315 (2001).
Properties
White crystalline solid. [α]D20 -55.9° (c = 0.5 in methanol). Soly (21°): 0.22 g/100 ml water.Derivative
Methanesulfonate salt.Nomenclature
CAS number: 149845-06-7
Saquinavir mesylate; Ro-31-8959/003; Fortovase (Roche); Invirase (Roche). C38H50N6O5.CH3SO3H; mol wt 766.95.
C 61.08%, H 7.10%, N 10.96%, O 16.69%, S 4.18%.
Therapeutic Category
Antiretroviral.
Keywords
HIV Protease Inhibitor; Antiretroviral; Protease Inhibitors