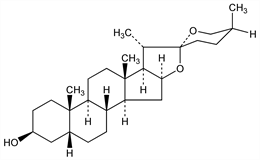
8380. Sarsasapogenin
Nomenclature
Description and references
Steroid sapogenin from Smilax ornata Hooker, fil., Liliaceae (Sarsaparilla): Power, Salway, J. Chem. Soc. 105, 201 (1914); Jacobs, Simpson, J. Biol. Chem. 105, 501 (1934); 109, 573 (1935). Partial structure: Hirschmann et al., J. Org. Chem. 20, 572 (1955). Stereochemistry: Taylor, Chem. Ind. (London) 1954, 1066; Wall, Serota, J. Am. Chem. Soc. 76, 2850 (1954); Callow, Massey-Beresford, J. Chem. Soc. 1957, 4482; Rosen et al., J. Am. Chem. Soc. 81, 1687 (1959). Reviews: Turner, The Steroidal Sapogenins Pub. no. 361, (Univ. Microfilms, Ann Arbor, Mich., 1941) 249 pp; Shabica, Sarsasapogenin and Related Compounds, Univ. Microfilms (Ann Arbor, Mich.), Pub. no. 549, 89 pp (1943); L. F. Fieser, M. Fieser, Steroids (Reinhold, New York, 1959) p 810. See also Smilagenin.