8401. Scilliroside
Nomenclature
CAS number: 507-60-8
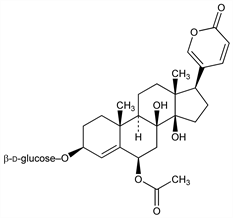
(3β,6β)-6-(Acetyloxy)-3-(β-d-glucopyranosyloxy)-8,14-dihydroxybufa-4,20,22-trienolide. C32H44O12; mol wt 620.68.
C 61.92%, H 7.15%, O 30.93%.
Description and references
Glycoside from red squill, the red variety of Urginea maritima (L.) Baker, Liliaceae: Stoll, Renz, Helv. Chim. Acta 25, 43 (1942); Wichtt, Fuchs, Arch. Pharm. 295, 361 (1962). Structure: Stoll et al., Helv. Chim. Acta 26, 648 (1943); von Wartburg, Renz, ibid. 42, 1620 (1959). Toxicity data: F. Dybing et al., Acta Pharmacol. Toxicol. 8, 391 (1952).
Properties
Long prisms from dilute methanol. The crystals are solvated and lose about 8% of their wt in high vacuum, but retain 1/2 mol H2O. For the hemihydrate, indistinct mp 168-170°, dec 200°. [α]D20 -59 to -60° (methanol). Absorption max (96% ethanol): 300 nm (log ε 3.73); (98% H2SO4): 295, 505 nm (E1%1cm 260, 315). Freely sol in the lower alcohols, in ethylene glycol, dioxane, glacial acetic acid. Slightly sol in water, acetone, chloroform, ethyl acetate. Practically insol in ether, petr ether. LD50 in mice (mg/kg): 0.471 s.c.; 0.440 orally (Dybing).Derivative
Tetraacetyl scilliroside.C40H52O16; mol wt 788.83.
C 60.90%, H 6.64%, O 32.45%.