8406. Scopolamine
Nomenclature
Description and references
Anticholinergic, tropane alkaloid isolated from Datura metel L., Scopola carniolica Jacq. and other Solanaceae. Constituent of impure duboisine from Duboisia myoporoides R. Br., pure duboisine is l-hyoscyamine, q.v. Isoln: A. Ladenburg, Ann. 206, 274 (1881); E. Schmidt, Arch. Pharm. 230, 207 (1892). Identity with hyoscine: O. Hesse, Ann. 271, 100 (1892); idem, J. Prakt. Chem. 66, 194 (1902). Absorption spectra: J. J. Dobbie, J. J. Fox, J. Chem. Soc. 103, 1194 (1913). Resolution of isomers and review of early literature: H. King, ibid. 1919, 476. Extraction procedure: F. Chemnitius, J. Prakt. Chem. 120, 221 (1928). Structural studies: J. Gadamer, F. Hammer, Arch. Pharm. 259, 122 (1921); K. Hess, O. Wahl, Ber. 55, 1979 (1922); R. Willst"atter, E. Berner, ibid. 56, 1079 (1923); W. Steffens, Arch. Pharm. 262, 205 (1924). Configuration: G. Fodor, Nature 170, 278 (1952); J. Meinwald, J. Chem. Soc. 1953, 712. Review of stereochemistry: G. Fodor, Tetrahedron 1, 86 (1957). Absolute configuration of tropic acid moiety: G. Fodor, G. Csepreghy, Tetrahedron Lett. 1959, 16; eidem, J. Chem. Soc. 1961, 3222. Synthesis of dl-form: G. Fodor et al., Chem. Ind. (London) 1956, 764; P. Dobo et al., J. Chem. Soc. 1959, 3461. Clinical evaluation in motion sickness: J. J. Brand, P. Whittingham, Lancet 2, 232 (1970); in peripheral vertigo: T. Rahko, P. Karma, J. Laryngol. Otol. 99, 653 (1985). Acute toxicity of the hydrobromide: Stockhaus, Wick, Arch. Int. Pharmacodyn. Ther. 180, 155 (1969). Review of CNS effects in humans: D. J. Safer, R. P. Allen, Biol. Psychiatry 3, 347-355 (1971); of use in anesthesia: L. E. Shutt, J. B. Bowles, Anaesthesia 34, 476-490 (1979); of pharmacology and clinical efficacy: S. P. Clissold, R. C. Heel, Drugs 29, 189-207 (1985). Comprehensive description: F. J. Muhtadi, M. M. A. Hassan, Anal. Profiles Drug Subs. 19, 477-551 (1990).
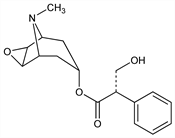
Properties
Viscous liquid. pKa 7.55-7.81. [α]D20 -28° (c = 2.7). Sol in 9.5 parts water at 15°. Forms a cryst monohydrate, mp 59°. Freely sol in hot water, in alcohol, ether, chloroform, acetone. Sparingly sol in benzene, petr ether. Easily hydrolyzed by acids or alkalies. Dec on standing.Derivative
Hydrobromide trihydrate.Nomenclature
Properties
Orthorhombic sphenoidal crystals from water, slightly efflorescent in dry air. mp 195° (after drying at 105° for 3 hours). [α]D25 -24 to -26° (c = 5, calculated on anhydrous basis). uv max (methanol) 246, 252, 258, 264 nm (A1%1cm 3.5, 4.0, 4.5, 3.0). pH of 0.05M soln 5.85. One gram dissolves in 1.5 ml water, 20 ml alcohol. Slightly sol in chloroform. Practically insol in ether. LD50 in rats (mg/kg): 3800 s.c. (Stockhaus, Wick).Derivative
Hydrochloride.Nomenclature
Properties
Crystals from acetone, mp 200°. Dihydrate, prisms from water, melts in water of crystn at 80°. Very sol in water and alcohol. pH of 0.05M soln 5.85.Derivative
Methyl bromide see Methscopolamine Bromide.Derivative
Methyl nitrate.Nomenclature
Properties
Crystals. Freely sol in water, dil alcohol; slightly sol in abs alcohol.Derivative
dl-Form.Nomenclature
Atroscine.Properties
Dihydrate, chisel-shaped prisms from ethanol + water, mp 38-40°. Monohydrate, efflorescent crystals, mp 55-57°. Anhydrous, long prisms, mp 82-83°. Very slightly sol in water; sol in alc, chloroform, ether, oils.Therapeutic Category
In treatment of motion sickness; antiemetic; antispasmodic; mydriatic; preanesthetic medicant.
Therapeutic Category (Veterinary)
Preanesthetic medicant.Keywords
Antiemetic; Mydriatic; Antispasmodic; Antimuscarinic