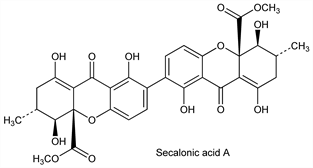
8418. Secalonic Acids
Description and references
Toxic mold metabolites which are diastereoisomeric dimeric hydroxanthone derivatives manifest as yellow pigments. First isolation studies: Stoll et al., Helv. Chim. Acta 35, 2022 (1952). Seven isomers have been isolated: secalonic acid A, secalonic acid B, and secalonic acid C from ergot: Aberhart et al., Tetrahedron 21, 1417 (1965); Franck et al., Ber. 99, 3842, 3875 (1966); secalonic acid D from Penicillium oxalicum: Steyn, Tetrahedron 26, 51 (1970); secalonic acid E from Phoma terrestris: Hansen, Howard et al., Chem. Commun. 1973, 464; secalonic acid F from Aspergillus aculeatus: R. Andersen et al., J. Org. Chem. 42, 352 (1977); secalonic acid G from Pyrenochaeta terrestris: I. Kurobane, L. C. Vining, Tetrahedron Lett. 1978, 4633. Secalonic acid A is enantiomeric with D; secalonic acid B is enantiomeric with E. Chromatographic separation of the ergot pigments: Franck, Angew. Chem. Int. Ed. 8, 251 (1969); structures: ApSimon et al., J. Chem. Soc. 1965, 4144. Revised structures of secalonic acids A, B, C, D: Hooper et al., Chem. Commun. 1971, 111; eidem, J. Chem. Soc. C 1971, 3580. Crystal and molecular structure of secalonic acid A: C. C. Howard et al., J. Chem. Soc. Perkin Trans. 1 1976, 1820. Identity of secalonic acid A with entothein: Yoshioka et al., Chem. Pharm. Bull. 16, 2090 (1968). Biosynthesis of secalonic acid A: I. Kurobane et al., Tetrahedron Lett. 1978, 1379. Review of secalonic acids and other ergochromes: Franck, Flasch in Fortschr. Chem. Org. Naturst. 30, 151-206 (1973).