8427. Selectride(R) (Aldrich)
Description and references
Group of stereoselective trialkyl borohydride reducing agents. Reduction of cycloalkenones: J. M. Fortunato, B. Ganem, J. Org. Chem. 41, 2194 (1976). Review of synthetic applications: S. Wittmann, B. Sch"onecker, J. Prakt. Chem. Chem.-Ztg. 338, 759-762 (1996).
Derivative
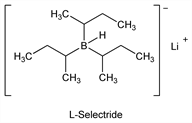
L-Selectride.Nomenclature
Description and references
Prepn and use in stereoselective reduction of cyclic ketones: H. C. Brown, S. Krishnamurthy, J. Am. Chem. Soc. 94, 7159 (1972). Synthetic use in reduction of cyclic anhydrides: M. A. Makhlouf, B. Rickborn, J. Org. Chem. 46, 4810 (1981); in cleavage of carbamates and O-demethylation of alkaloids: A. Coop et al., ibid. 63, 4392 (1998).
Derivative
K-Selectride.Nomenclature
Description and references
Prepn: C. A. Brown, J. Am. Chem. Soc. 95, 4100 (1973); idem, Inorg. Synth. 17, 26 (1977). Synthetic use in conjugate reduction and reductive alkylation of α,β-unsaturated cyclohexenones: B. Ganem, J. Org. Chem. 40, 146 (1975); in reduction of diketo steroids: D. M. Tal et al., Tetrahedron 40, 851 (1984).