8428. Selegiline
Nomenclature
Description and references
Monoamine oxidase-B inhibitor related structurally to pargyline, q.v. Prepn of racemate: FR 1368136; GB 1031425 (1964, 1966 both to Chinoin); of (-)-form: NL 6605956; GB 1153578 (1966, 1969 both to Chinoin); J. S. Fowler, J. Org. Chem. 42, 2637 (1977) DOI PubMed. Pharmacology and toxicology of racemate: J. Knoll et al., Arch. Int. Pharmacodyn. 155, 154 (1965) PubMed; of isomers: K. Magyar et al., Acta Physiol. Acad. Sci. Hung. 32, 377 (1967) PubMed. Review of pharmacology: E. H. Heinonen, R. Lammintausta, Acta Neurol. Scand. Suppl. 136, 44-59 (1991) PubMed; of efficacy in Parkinson's disease: D. W. Robin, Am. J. Med. Sci. 302, 392-395 (1991) DOI PubMed; K. L. Poston, C. Waters, Expert Opin. Pharmacother. 8, 2615-2624 (2007) DOI PubMed. Review of veterinary use in canine cognitive dysfunction: W. W. Ruehl, B. L. Hart in Psychopharmacology of Animal Behavior Disorders, N. H. Dodman, L. Shuster, Eds. (Blackwell Sci., Malden, Mass., 1998) pp 283-304. Review of transdermal selegiline in major depressive disorder: D. S. Robinson, J. D. Amsterdam, J. Affect. Disord. 105, 15-23 (2008) DOI PubMed.
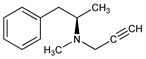
Properties
Oil, bp0.8 92-93°. nD20 1.5180.Derivative
Hydrochloride.Nomenclature
Properties
Crystals, mp 141-142°. [α]D25 -10.8° (c = 6.48 in water). Freely sol in water, chloroform, methanol. LD50 in rats (mg/kg): 81 i.v., 280 s.c. (Magyar).Derivative
(±)-Form.Nomenclature
Properties
Oil, bp5 104-110°. nD20 1.5229.Derivative
(±)-Form hydrochloride.Nomenclature
Properties
Crystals from ethanol + ether, mp 131-131.5°. LD50 in rats (mg/kg): 63 i.v., 126 s.c., 385 orally (Knoll).Therapeutic Category
Antiparkinsonian; antidepressent.
Therapeutic Category (Veterinary)
In treatment of canine cognitive dysfunction syndrome.Keywords
Antiparkinsonian; Monoamine Oxidase Inhibitor; Antidepressant