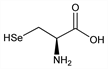
8440. Selenocysteine
Nomenclature
Description and references
Naturally occurring amino acid found in the active site of certain prokaryotic and eukaryotic enzymes. Highly oxidizable making synthesis difficult. Easily prepared from its oxidized form selenocystine. Prepn of dl-selenocystine: A. Fredga, Sven. Kem. Tidskr. 48, 160 (1936); E. P. Painter, J. Chem. Soc. 69, 229 (1947); of l-form: H. Tanaka, K. Soda, Methods Enzymol. 143, 240 (1987). Reduction of selenocystine and separation of l-selenocysteine: J. N. Burnell et al., J. Inorg. Biochem. 12, 343 (1980). Chemical properties: R. E. Huber, R. S. Criddle, Arch. Biochem. Biophys. 122, 164 (1967). Determn by HPLC/MS: H. E. Ganther et al., Methods Enzymol. 107, 582 (1984); colorimetric determn: N. Esaki, K. Soda, ibid. 143, 148 (1987). Identification of Se as a component in protein: S.-H. Oh et al., Biochemistry 13, 1825 (1974). Characterization of selenocysteine as the Se containing amino acid moiety in various enzymes: J. E. Cone et al., Proc. Natl. Acad. Sci. USA 73, 2659 (1976); J. B. Jones et al., Arch. Biochem. Biophys. 195, 255 (1979); presence in the catalytic site of mammalian glutathione peroxidase: J. W. Forstrom et al., Biochemistry 17, 2639 (1978); J. J. Zabrowski et al., Biochem. Biophys. Res. Commun. 84, 248 (1978). Metabolism in animals: H. Tanaka et al., Curr. Top. Cell. Regul. 27, 487 (1985). Identification of selenocysteine-specific tRNA: W. C. Hawkes et al., Biochim. Biophys. Acta 699, 183 (1982); W. Leinfelder et al., Nature 331, 723 (1988). Review: T. C. Stadtman, FASEB J. 1, 375-379 (1987).