8455. Sennosides
Nomenclature
Colonorm (Mundipharma); Glysennid (Dorsey); Nytilax (Rohto); Pursennid (Novartis).Description and references
Anthraquinone glucosides found in senna in equal amts and in the rhubarbs where sennoside A predominates. Isoln from Tinnevelly senna (Cassia angustifolia Vahl, Leguminosae): Stoll et al., Helv. Chim. Acta 32, 1892 (1949); GB 804232 (1958 to Byk-Gulden); Khorana, Sanghavi, J. Pharm. Sci. 53, 110 (1964); Menssen et al., US 3517269 (1970 to Nattermann & Cie). Isoln from the rhyzome of Rheum palmatum L.: Zwaving, Planta Med. 13, 474 (1965). Isoln of sennoside A from rhubarb: Miyamoto et al., Yakugaku Zasshi 87, 1040 (1967). Sennosides B, C, D, E, and F have also been isolated from rhubarb, see Oshio et al., Chem. Pharm. Bull. 22, 823 (1974). Structure: Stoll et al., Helv. Chim. Acta 33, 313 (1950). Sennoside A is built up from the dextrorotatory aglucon sennidin A and d-glucose. Sennoside B is built up from the intramolecularly compensated meso-sennidin B and d-glucose. Review: Stoll, Becker, Fortschr. Chem. Org. Naturst. 7, 248 (1950). Transport and mechanism of action studies: Dobbs et al., Farmaco Ed. Sci. 30, 147 (1975).
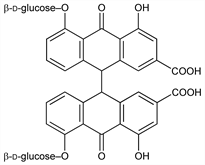
Derivative
Sennoside A.Nomenclature
Properties
Rectangular yellow plates from dil acetone, dec 200-240°. [α]D20 -164° (c = 0.1 in 60% acetone); [α]D20 -147° (c = 0.1 in 70% acetone); [α]D20 -24° (c = 0.2 in 70% dioxane). Insol in water, benzene, ether, chloroform. Sparingly sol in methanol, Carbitol, acetone, dioxane. The soly increases tremendously in water-miscible organic solvents contg an optimum of 30% w/w of water. Sol in aq solns of sodium bicarbonate. Sennoside A can be slowly isomerized to B in NaHCO3 soln at 80°.Derivative
Sennoside B.Nomenclature
Properties
Light yellow prisms from dil acetone; fine needles from water, dec 180-186°. [α]D20 -100° (c = 0.2 in 70% acetone); [α]D20 -67° (c = 0.4 in 70% dioxane). uv max (0.5% NaHCO3): 270, 308, 355 nm. Has the same soly characteristics as sennoside A, but is more sol. Can be recrystallized from large amounts of hot water. Keep protected from air and light.Therapeutic Category
Cathartic.
Keywords
Laxative/Cathartic