8456. Seocalcitol
Nomenclature
Description and references
Antiproliferative vitamin D analog with reduced effects on calcium metabolism. Prepn: E. Binderup, M. J. Calverley, WO 9100855; US 5190935 (1991, 1993 both to Leo Pharm.). LC-MS-MS determn in serum: A.-M. Kissmeyer et al., J. Chromatogr. B 740, 117 (2000). Pharmacokinetics and metabolism: A.-M. Kissmeyer, J. T. Mortensen, Xenobiotica 30, 815 (2000). Mechanism of action study: G. A. DeMasters et al., J. Steroid Biochem. Mol. Biol. 92, 365 (2004). Clinical evaluation in pancreatic cancer: T. R. J. Evans et al., Br. J. Cancer 86, 680 (2002); in hepatocellular carcinoma: K. Dalhoff et al., Br. J. Cancer 89, 252 (2003). Review of pharmacology and clinical experience: C. M. Hansen et al., Curr. Pharm. Des. 6, 803-828 (2000).
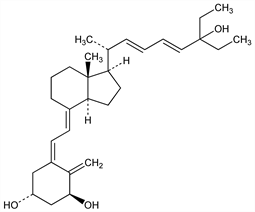
Properties
Needles from methyl formate-hexane, mp 123-125°. uv max: 232, 264 nm (ε 43745, 18060).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic