8541. Sinapine
Nomenclature
CAS number: 18696-26-9
2-[[3-(4-Hydroxy-3,5-dimethoxyphenyl)-1-oxo-2-propenyl]oxy]-N,N,N-trimethylethanaminium; sinapic acid choline ester. [C16H24NO5]+.
Description and references
From black mustard seeds, the seeds of Brassica nigra Koch, Cruciferae. Extraction procedure: Remsen, Coale, Am. Chem. J. 6, 52 (1884). Synthesis: Sp"ath, Monatsh. Chem. 41, 271 (1920).
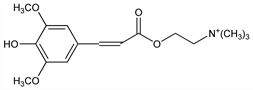
Derivative
Chloride.(C16H24NO5)Cl; mol wt 345.82.
C 55.57%, H 7.00%, N 4.05%, O 23.13%, Cl 10.25%.
Properties
Does not crystallize, very sol in water, alcohol.Derivative
Bromide trihydrate.(C16H24NO5)Br.3H2O; mol wt 444.32.
C 43.25%, H 6.81%, N 3.15%, O 28.81%, Br 17.98%.
Properties
Needles, mp 92°; mp 107-115° when anhydr, very sol in water.Derivative
Iodide trihydrate.(C16H24NO5)I.3H2O; mol wt 491.32.
C 39.11%, H 6.15%, N 2.85%, O 26.05%, I 25.83%.
Properties
Crystals, becomes anhydr over H2SO4, mp 179° (anhydr). Sparingly sol in water.Derivative
Acid sulfate trihydrate.(C16H24NO5)HSO4.3H2O; mol wt 461.48.
C 41.64%, H 6.77%, N 3.04%, O 41.60%, S 6.95%.
Properties
Tablets, dec 127°; dec 187° when anhydr. Freely sol in water, hot alcohol; very sparingly sol in ether.Derivative
Neutral sulfate pentahydrate.(C16H24NO5)2SO4.5H2O; mol wt 806.87.
C 47.63%, H 7.25%, N 3.47%, O 37.68%, S 3.97%.