8550. Sisomicin
Nomenclature
Description and references
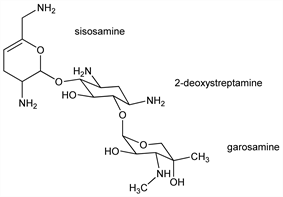
Gentamicin-like aminoglycoside antibiotic produced by Micromonospora inyoesis (NRRL 3292): M. J. Weinstein et al. DE 1932309; eidem, US 3832286 (1970, 1974 both to Schering); eidem, J. Antibiot. 23, 551 (1970). Isoln: G. H. Wagman et al., ibid. 555. Chemical and physical studies: Cooper et al., Chem. Commun. 1971, 285, 924; Kugelman et al., J. Antibiot. 26, 394 (1973). Structure consists of 2-deoxystreptamine, q.v., linked to two saccharide units, garosamine and sisosamine: Keiman et al., J. Org. Chem. 39, 1451 (1974). Synthesis: D. H. Davies et al., J. Med. Chem. 21, 189 (1978). Synthesis of sisosamine derivs: Cleophax et al., Chem. Commun. 1975, 11. Has broad spectrum antibiotic activity: Waitz et al., J. Antibiot. 23, 559 (1970). Biotransformation of sisomicin to gentamicin: B. K. Lee et al., Antimicrob. Agents Chemother. 12, 335 (1977). Clinical studies: R. Lorber et al., Clin. Ther. 4, 263 (1981); J. P. Sculier et al., J. Antimicrob. Chemother. 9, 63 (1982). Review: P. Noone, Drugs 27, 548-578 (1984).
Derivative
Monohydrate.Properties
Needles from ethanol, mp 198-201°. [α]D26 +189° (c = 0.3).Derivative
Penta-N-acetyl deriv.Properties
Amorphous white powder from methanol-ethyl ether, mp 188-198° (dec). [α]D26 +200° (c = 0.3).Derivative
Sulfate.Nomenclature
Properties
LD50 in mice (mg/kg): 34 i.v., 221 i.p., 288 s.c. (Weinstein).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Aminoglycosides