8552. Sitagliptin
Nomenclature
Description and references
Selective inhibitor of dipeptidyl peptidase IV (DPP-IV). Prepn: S. D. Edmondson et al., WO 03004498; eidem, US 6699871 (2003, 2004 both to Merck & Co.); and enzyme inhibitory profile: D. Kim et al., J. Med. Chem. 48, 141 (2005). Improved process: K. B. Hansen et al., Org. Process Res. Dev. 9, 634 (2005). Discovery and characterization: N. A. Thornberry, A. E. Weber, Curr. Top. Med. Chem. 7, 557 (2007). HTLC determn in plasma: W. Zeng et al., Rapid Commun. Mass Spectrom. 20, 1169 (2006). Clinical pharmacokinetics and pharmacodynamics: G. A. Herman et al., Clin. Pharmacol. Ther. 78, 675 (2005). Review: C. F. Deacon, Curr. Opin. Investig. Drugs 6, 419-426 (2005). Review of pharmacology and clinical experience: M. Choy, S. Lam, Cardiology Rev. 15, 264-271 (2007).
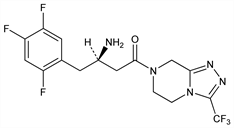
Properties
Viscous oil.Derivative
Monophosphate monohydrate.Nomenclature
Properties
White to off-white, crystalline, nonhygroscopic powder, mp 215-217°. [α]D -74.4° (c = 1.0 in water). Sol in water, DMF; slightly sol in methanol; very slightly sol in ethanol, acetone, acetonitrile. Insol in isopropanol, isopropyl acetate.Therapeutic Category
Antidiabetic.
Keywords
Antidiabetic; Dipeptidyl Peptidase IV Inhibitor