8553. Sitamaquine
Nomenclature
Description and references
8-Aminoquinolone; analog of the antimalarial agent primaquine, q.v. Prepn: K. N. Campbell et al., J. Am. Chem. Soc. 68, 1556 (1946); idem, US 2508937 (1950 to USA, as represented by the Secy. of War). Antileishmanial activity in hamsters: K. E. Kinnamon et al., Am. J. Trop. Med. Hyg. 27, 751 (1978). HPLC determn in plasma: J. C. Anders et al., J. Chromatogr. 311, 117 (1984). Clinical evaluation in AIDS-associated Pneumocystis carinii pneumonia: B. G. Petty et al., J. Acquir. Immune Defic. Syndr. 21, 26 (1999); in visceral leishmaniasis: R. Dietze et al., Am. J. Trop. Med. Hyg. 65, 685 (2001). Ex vivo use in prevention of transfusion-associated Chagas' disease: H. Moraes-Souza et al., Rev. Soc. Bras. Med. Trop. 35, 563 (2002). Review of clinical development: C. Yeates, Curr. Opin. Investig. Drugs 3, 1446-1452 (2002); H. Sangraula et al., J. Assoc. Physicians India 51, 686-690 (2003).
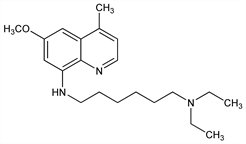
Properties
bp0.05 190-200°.Derivative
Dihydrochloride.Nomenclature
Properties
Yellow crystals from propanol, mp 179-180°.Therapeutic Category
Antiprotozoal (Leishmania).
Keywords
Antiprotozoal (Leishmania)