8705. Solanine
Nomenclature
Description and references
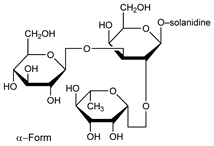
From Solanum species, especially from S. tuberosum L. (potato), S. nigrum L. (woody nightshade), and Lycopersicon esculentum Mill., Solanaceae (tomato). Fresh potato sprouts contain about 0.04%. Extraction procedure: Soltys, Wallenfels, Ber. 69, 811 (1936). Separated by chromatography into six components: α-, β-, γ-solanine and α-, β-, γ-chaconine. Main constituent α-solanine: Kuhn, Lw, Angew. Chem. 66, 639 (1954); Kuhn et al., Ber. 88, 1492 (1955); Paseshnichenko, Guseva, Biokhimiya 21, 585 (1956); eidem, ibid. 22, 843 (1957). Pharmacology: Nishie et al., Toxicol. Appl. Pharmacol. 19, 81 (1971). Toxicity study: B. C. Patil et al., Food Cosmet. Toxicol. 10, 395 (1972); and teratogenicity study: S. Chaube, C. A. Swinyard, Toxicol. Appl. Pharmacol. 36, 227 (1976).