8711. Solasulfone
Nomenclature
CAS number: 133-65-3
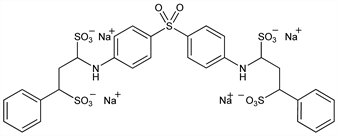
1,1′-[Sulfonylbis(4,1-phenyleneimino)]bis[3-phenyl-1,3-propanedisulfonic
acid] tetrasodium salt; di(γ-phenylpropylamino)-4,4′-diphenylsulfone α,α′,γ,γ′-sodium
tetrasulfonate; tetrasodium 4,4′-bis(γ-phenylpropylamino)diphenylsulfone α,γ,α′,γ′-tetrasulfonate; phenopryldiasulfone sodium; solapsone; RP-3668; Cimedone (Specia); Sulphetrone (Burroughs Wellcome). C30H28N2Na4O14S5; mol wt 892.83.
C 40.36%, H 3.16%, N 3.14%, Na 10.30%, O 25.09%, S 17.96%.
Description and references
Prepared by reacting 4,4′-diaminodiphenyl sulfone with more than 2 mols cinnamic aldehyde in the cold and treating the resulting 4,4′-dicinnamylideneaminodiphenyl sulfone with NaHSO3 soln: Gray, Henry, GB 491265 (1938 to Wellcome Foundation); Buttle et al., Biochem. J. 32, 1101 (1938). Improved process: Henry, Gray, GB 562216 (1944 to Wellcome Foundation). Properties and estimation: Brownlee et al., Br. J. Pharmacol. 3, 15 (1948).
Properties
Crystals contg 5-7% H2O from alc. Becomes anhydr at 110° in vacuo. Exceedingly sol in cold water, giving a neutral soln. 20% and 40% (w/v) aq solns are stable when neutral or slightly alkaline and may be autoclaved. Practically insol in cold alc. Solns are incompatible with acids. Absorption spectrum of dye obtained by diazotization and coupling to N-(1-naphthyl)ethylenediamine hydrochloride: Brownlee, loc. cit.Therapeutic Category
Antibacterial (leprostatic).
Keywords
Antibacterial (Synthetic); Sulfones; Antibacterial (Leprostatic)