8752. Spiramycin
Nomenclature
Description and references
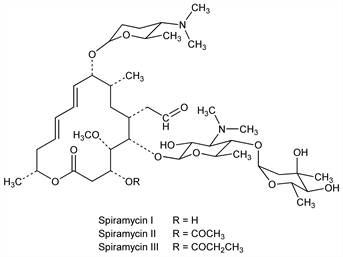
Antibiotic substance classified in the erythromycin-carbomycin group and produced by Streptomyces ambofaciens from soil of northern France: Cosar et al., C. R. Seances Soc. Biol. Ses Fil. 234, 1498 (1952); Pinnert-Sindico et al., Antibiot. Annu. 1954-1955, 724; Ninet, Verrier, US 2943023 (1960 to Rhne-Poulenc), see also US 3000785 (1961 to Rhne-Poulenc). Antibacterial activity and toxicity: H. Sous et al., Arzneim.-Forsch. 8, 386 (1958). Separation into 3 components named spiramycin I, II and III: Preud'homme, Charpentier, US 2978380 and US 3011947 (1961 to Rhne-Poulenc). Structure: Kuehne, Benson, J. Am. Chem. Soc. 87, 4660 (1965). Revised structure: Omura et al., ibid. 91, 3401 (1969); Mitscher et al., J. Antibiot. 26, 55 (1973). Revised configuration at C-9: Freiberg et al., J. Org. Chem. 39, 2474 (1974). Symposium on pharmacology, antibacterial spectrum, and clinical efficacy: J. Antimicrob. Chemother. 22, Suppl. B, 1-213 (1988).
Properties
Amorphous base, slightly sol in water. [α]D20 80° (methanol). uv max (ethanol): 231 nm. Sol in most organic solvents. Active on gram-positive bacteria and rickettsiae. Cross resistance between microorganisms resistant to erythromycin and carbomycin. LD50 in rats (mg/kg): 9400 orally; 1000 s.c.; 170 i.v. (Sous).Derivative
Embonate.Nomenclature
Spira 200 (RMB).Derivative
Hexanedioate.Nomenclature
Spiramycin adipate; Stomamycin (Chassot); Suanovil (Biokema).Derivative
Spiramycin I.Nomenclature
Properties
Crystals, mp 134-137°. [α]D20 96°.Derivative
Spiramycin I triacetate.Properties
Crystals, mp 140-142°. [α]D20 92.5°.Derivative
Spiramycin II.Nomenclature
Properties
Crystals, mp 130-133°. [α]D20 86°.Derivative
Spiramycin II diacetate.Properties
Crystals from cyclohexane, mp 156-160°. [α]D20 98.4°.Derivative
Spiramycin III.Nomenclature
Properties
Crystals, mp 128-131°. [α]D20 83°.Derivative
Spiramycin III diacetate.Properties
Crystals from cyclohexane, mp 140-142°. [α]D20 90.4°.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial; growth promotant.Keywords
Antibacterial (Antibiotics); Macrolides