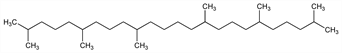
8767. Squalane
Nomenclature
CAS number: 111-01-3
2,6,10,15,19,23-Hexamethyltetracosane; perhydrosqualene; dodecahydrosqualene; spinacane; Cosbiol (Laserson & Sabetay); Robane (Robeco); Wax-O-Sol (Life Sci.). C30H62; mol wt 422.81.
C 85.22%, H 14.78%.
Description and references
Prepn by complete hydrogenation of squalene, q.v.: Chapman, J. Chem. Soc. 123, 770 (1923); Heilbron et al., ibid. 1926, 3135. Commercial grades are obtained by direct hydrogenation of shark liver oil and may contain some batyl alcohol: Tsujimoto, Chem. Umschau Fette 34, 256 (1927), C.A. 21, 4081 (1927). Synthesis: J. W. Scott, D. Valentine, Org. Prep. Proced. Int. 12, 7 (1980); T. Mandai et al., Tetrahedron Lett. 22, 763 (1981).